すべての画像(2)
About This Item
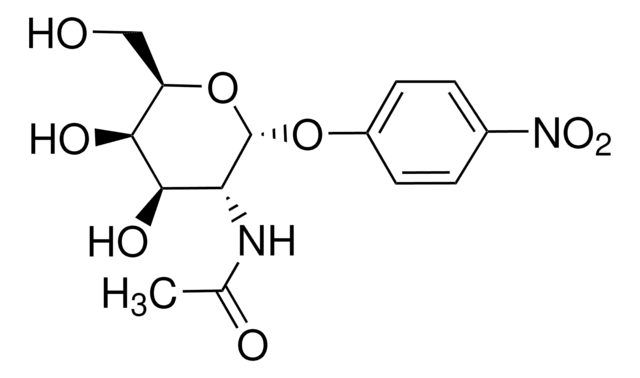
実験式(ヒル表記法):
C5H10N2O · HCl
CAS番号:
分子量:
150.61
Beilstein:
3693546
EC Number:
MDL番号:
UNSPSCコード:
12352200
PubChem Substance ID:
NACRES:
NA.26
おすすめの製品
フォーム
powder
SMILES記法
Cl.NC(=O)[C@@H]1CCCN1
InChI
1S/C5H10N2O.ClH/c6-5(8)4-2-1-3-7-4;/h4,7H,1-3H2,(H2,6,8);1H/t4-;/m0./s1
InChI Key
CSKSDAVTCKIENY-WCCKRBBISA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
Eyeshields, Gloves, type N95 (US)
Martin G Schmid et al.
Analytical and bioanalytical chemistry, 400(8), 2305-2316 (2011-02-15)
This article gives a short overview of the application of the principle of chiral ligand-exchange in HPLC, CE, and CEC. Since its introduction by Davankov, more than thousand articles have appeared in this field. Citing all these papers would extend
Hizuru Nakajima et al.
Analytical sciences : the international journal of the Japan Society for Analytical Chemistry, 21(1), 67-71 (2005-01-29)
The chiral separation of amino acid derivatives by ligand-exchange electrophoresis in a microchannel chip was performed for the first time. A Cu(II) complex with L-prolinamide was used as a chiral selector. The migration behaviors of eleven NBD-DL-amino acids were investigated
Li Qi et al.
Journal of separation science, 32(18), 3209-3214 (2009-08-26)
A novel method of chiral ligand-exchange CE was developed with L-amino acylamides as a chiral ligand and zinc(II) as a central ion. It has been demonstrated that these chiral complexes, such as Zn(II)-L-alaninamide, Zn(II)-L-prolinamide, and Zn(II)-L-phenylalaninamide, are suitable for use
Patrycja Puchalska et al.
Electrophoresis, 31(9), 1517-1520 (2010-04-28)
A new chiral stationary phase based on continuous bed (CB) technology using L-prolinamide as a chiral selector was prepared. Its ability for enantioseparation of amino acids and alpha-hydroxy acids by ligand-exchange CEC was compared with that of a CB containing
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)