すべての画像(1)
About This Item
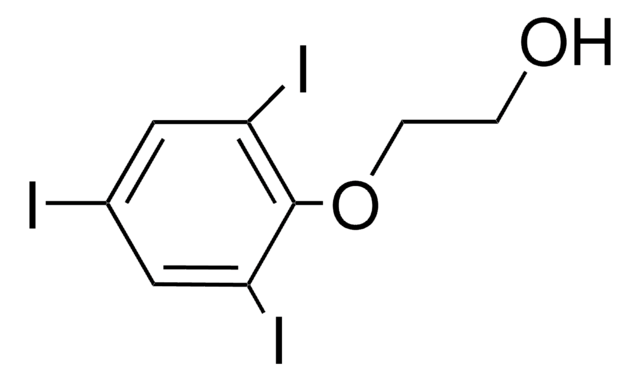
化学式:
HOC6H(I)3CH2CH(C2H5)CO2H
CAS番号:
分子量:
571.92
MDL番号:
UNSPSCコード:
12352100
PubChem Substance ID:
NACRES:
NA.22
おすすめの製品
アッセイ
97%
形状
solid
mp
146-148 °C (lit.)
SMILES記法
CCC(Cc1c(I)cc(I)c(O)c1I)C(O)=O
InChI
1S/C11H11I3O3/c1-2-5(11(16)17)3-6-7(12)4-8(13)10(15)9(6)14/h4-5,15H,2-3H2,1H3,(H,16,17)
InChI Key
GOIQOQCNFWYSTQ-UHFFFAOYSA-N
関連するカテゴリー
詳細
α-Ethyl-3-hydroxy-2,4,6-triiodohydrocinnamic acid (Iophenoxic acid ) is an organic, iodine-containing compound. Iophenoxic acid is an iodinated radiocontrast agent, its clinical use has been withdrawn due to its exceptionally long half-life in the body, since it has high-affinity binding to human serum albumin (HSA). Structural basis of its interaction with HSA has been evaluated.
アプリケーション
α-Ethyl-3-hydroxy-2,4,6-triiodohydrocinnamic acid (iophenoxic acid ) was used as marker in “bait acceptance” studies conducted on various animal species. It was used in the direct quantitation of iophenoxic acid in porcine serum samples by a liquid chromatographic-electrospray ionization mass spectrometric technique.
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
ターゲットの組織
Respiratory system
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Gloves
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
労働安全衛生法名称等を表示すべき危険物及び有害物
名称等を表示すべき危険物及び有害物
労働安全衛生法名称等を通知すべき危険物及び有害物
名称等を通知すべき危険物及び有害物
Jan Code
361046-VAR:
361046-5G:4548173993652
361046-BULK:
361046-1G:
試験成績書(COA)
製品のロット番号・バッチ番号を入力して、試験成績書(COA) を検索できます。ロット番号・バッチ番号は、製品ラベルに「Lot」または「Batch」に続いて記載されています。
Carlo Bertucci et al.
Farmaco (Societa chimica italiana : 1989), 58(9), 901-908 (2003-09-19)
The binding of two cholecystographic agents to human serum albumin (HSA) was evaluated by means of two different complementary methodologies. In particular, the inhibition of drug HSA binding caused by iopanoic- and iophenoxic-acid was investigated by circular dichroism (CD) and
C T Eason et al.
Xenobiotica; the fate of foreign compounds in biological systems, 22(2), 185-189 (1992-02-01)
1. The comparative plasma pharmacokinetics of two organic iodine-containing compounds were evaluated in the goat for their suitability as markers in wildlife studies. 2. After oral administration of a single dose, the plasma elimination half-life for iopanoic acid was considerably
A Jones
Journal of chromatography. B, Biomedical applications, 654(2), 293-296 (1994-04-01)
Iophenoxic acid (IPA), a marker used to investigate the feeding behaviour of bait-consuming animals has previously been indirectly determined by measuring protein-bound iodine levels in serum or plasma. For the first time a method is reported for the direct determination
Ali J Ryan et al.
BMC structural biology, 11, 18-18 (2011-04-20)
Iophenoxic acid is an iodinated radiocontrast agent that was withdrawn from clinical use because of its exceptionally long half-life in the body, which was due in part to its high-affinity binding to human serum albumin (HSA). It was replaced by
J Hadidian et al.
Journal of wildlife diseases, 25(1), 1-9 (1989-01-01)
In summer 1986, a study was conducted to evaluate raccoon (Procyon lotor) acceptance of oral baits that could be used for rabies vaccination. One thousand wax-coated sponge bait cubes were filled with 5 mg of a seromarker (iophenoxic acid), placed
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)