D194255
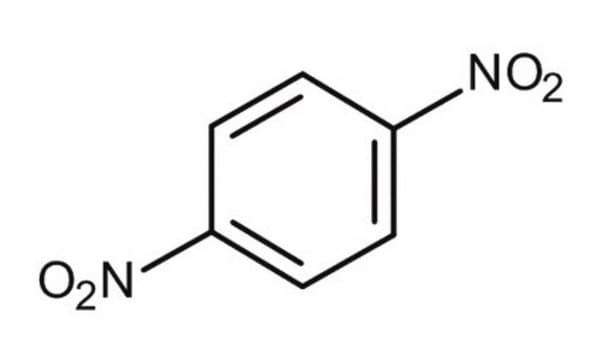
1,3-Dinitrobenzene
97% anhydrous basis
Sinónimos:
1,3-Dinitrobenzene, DNB, Meta-dinitrobenzene, m-Dinitrobenzene
About This Item
Productos recomendados
Quality Level
assay
97% anhydrous basis
form
solid
impurities
≤10.0% H2O
bp
297 °C (lit.)
mp
84-86 °C (lit.)
density
1.368 g/mL at 25 °C (lit.)
SMILES string
[O-][N+](=O)c1cccc(c1)[N+]([O-])=O
InChI
1S/C6H4N2O4/c9-7(10)5-2-1-3-6(4-5)8(11)12/h1-4H
InChI key
WDCYWAQPCXBPJA-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- 2′,6′- dinitrobiphenyl-4-ol and 1-nitrodibenzofuran via copper-catalyzed regioselective cross-coupling reaction with 4-iodophenol and 2-iodophenol respectively.
- 1H-indazole derivatives by reacting with various N-tosylhydrazones in the presence of a base catalyst.
- 3-(1H-Tetrazol-1-yl)benzenamine by reacting with triethyl orthoformate and NaN3 in the presence of a three-functional redox catalytic system.
signalword
Danger
Hazard Classifications
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
302.0 °F - closed cup
flash_point_c
150 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico