126632
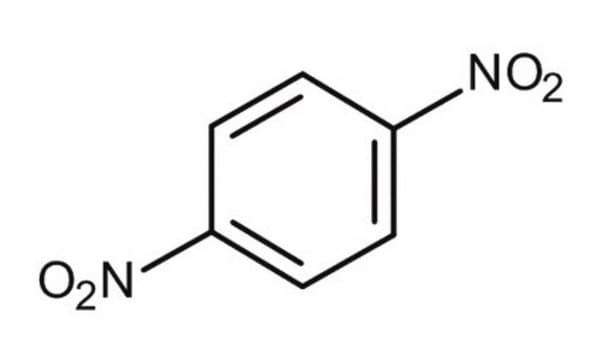
1,2-Dinitrobenzene
97%
Sinónimos:
o-Dinitrobenzene
About This Item
Productos recomendados
Nivel de calidad
Ensayo
97%
Formulario
solid
bp
319 °C/773 mmHg (lit.)
mp
114-117 °C (lit.)
grupo funcional
nitro
cadena SMILES
[O-][N+](=O)c1ccccc1[N+]([O-])=O
InChI
1S/C6H4N2O4/c9-7(10)5-3-1-2-4-6(5)8(11)12/h1-4H
Clave InChI
IZUKQUVSCNEFMJ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
- A highly divergent Pictet-Spengler approach for pyrrolo quinoxalines from aryl amine using 1,2-dinitrobenzene as an oxidant: Discusses the utility of 1,2-dinitrobenzene as an oxidant in organic synthesis, relevant for developing novel pharmaceuticals (Pardeshi et al., 2019).
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - STOT RE 2
Código de clase de almacenamiento
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
302.0 °F - closed cup
Punto de inflamabilidad (°C)
150 °C - closed cup
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico