126179
9,10-Dihydroanthracene
97%
Sinónimos:
Dihydroanthracene
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C14H12
Número de CAS:
Peso molecular:
180.25
Beilstein/REAXYS Number:
1364575
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
97%
form
solid
bp
312 °C (lit.)
mp
103-107 °C (lit.)
density
0.88 g/mL at 25 °C (lit.)
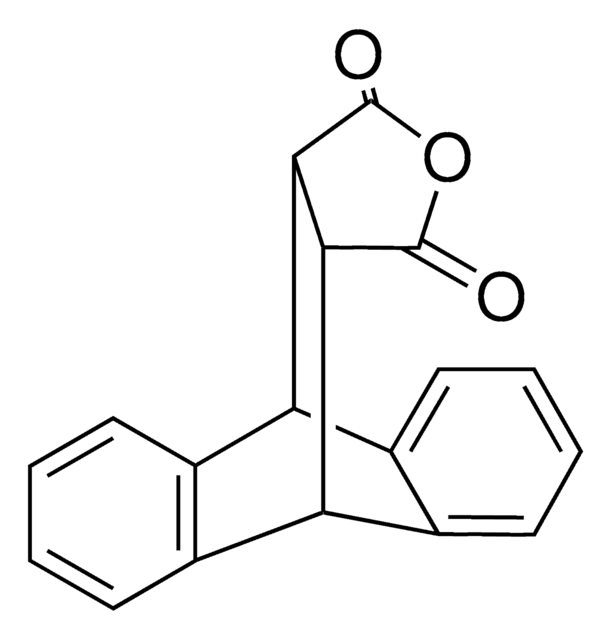
SMILES string
C1c2ccccc2Cc3ccccc13
InChI
1S/C14H12/c1-2-6-12-10-14-8-4-3-7-13(14)9-11(12)5-1/h1-8H,9-10H2
InChI key
WPDAVTSOEQEGMS-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
9,10-Dihydroanthracene causes the transfer hydrogenation of C60 and C70 in the presence of [7H]benzanthrene catalyst. It was oxidatively aromatized to the corresponding anthracene in the presence of molecular oxygen as an oxidant and activated carbon as a promoter in xylene.
Application
9,10-Dihydroanthracene(DHA) has been used in a study to assess the hydrogen abstraction capability of valence-delocalized iron complex with DHA in MeCN.
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Natsuki Nakamichi et al.
The Journal of organic chemistry, 68(21), 8272-8273 (2003-10-11)
Substituted 9,10-dihydroanthracenes were oxidatively aromatized to the corresponding anthracenes effectively by using molecular oxygen as an oxidant and activated carbon (Darco KB, Aldrich, Inc.) as a promoter in xylene.
[7H] Benzanthrone, a catalyst for the transfer hydrogenation of C60 and C70 by 9, 10-dihydroanthracene.
Gerst M, et al.
Tetrahedron Letters, 34(48), 7729-7732 (1993)
Pengfei Sun et al.
Small (Weinheim an der Bergstrasse, Germany), 16(3), e1906462-e1906462 (2019-12-24)
Lithium primary batteries are still widely used in military, aerospace, medical, and civilian applications despite the omnipresence of rechargeable Li-ion batteries. However, these current primary chemistries are exclusively based on inorganic materials with high cost, low energy density or severe
Nirupama Singh et al.
Journal of inorganic biochemistry, 197, 110674-110674 (2019-05-21)
Developing coordination complexes of earth abundant metals that can perform substrate oxidations under benign conditions is an ongoing challenge. Herein, the reactivity of two mononuclear Cu-complexes toward the oxidant H2O2 is reported. Both complexes displayed ligand oxidation upon reaction with
Genqiang Xue et al.
Proceedings of the National Academy of Sciences of the United States of America, 104(52), 20713-20718 (2007-12-21)
Intermediate Q, the methane-oxidizing species of soluble methane monooxygenase, is proposed to have an [Fe(IV)(2)(mu-O)(2)] diamond core. In an effort to obtain a synthetic precedent for such a core, bulk electrolysis at 900 mV (versus Fc(+/0)) has been performed in
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico