所有图片(4)
About This Item
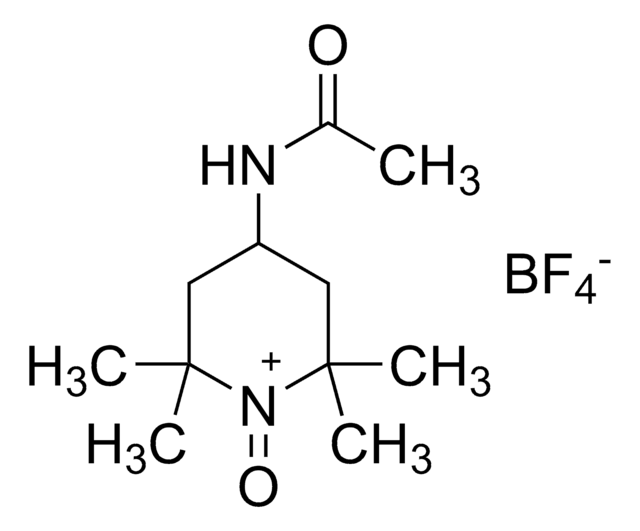
经验公式(希尔记法):
C11H21N2O2
CAS号:
分子量:
213.30
Beilstein:
3546225
EC 号:
MDL编号:
UNSPSC代码:
12352005
PubChem化学物质编号:
NACRES:
NA.22
推荐产品
等级
purum
质量水平
方案
≥98.0% (HPLC)
环保替代产品得分
old score: 9
new score: 8
Find out more about DOZN™ Scoring
环保替代产品特性
Waste Prevention
Safer Solvents and Auxiliaries
Inherently Safer Chemistry for Accident Prevention
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
143-145 °C (lit.)
143-146 °C
官能团
amide
环保替代产品分类
储存温度
2-8°C
SMILES字符串
CC(=O)NC1CC(C)(C)N([O])C(C)(C)C1
InChI
1S/C11H21N2O2/c1-8(14)12-9-6-10(2,3)13(15)11(4,5)7-9/h9H,6-7H2,1-5H3,(H,12,14)
InChI key
UXBLSWOMIHTQPH-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
相关类别
一般描述
4-Acetamido-2,2,6,6-tetramethylpiperidine 1-oxyl undergoes one-electron oxidation and reduction reactions. Reaction products were analyzed by 1H, 13C and 15N NMR spectral data.
We are committed to bringing you Greener Alternative Products, which adhere to one of the four categories of Greener Alternatives . This product belongs to category of Re-engineered products, showing key improvements in Green Chemistry Principles “Waste Prevention”, “Safer Solvents and Auxiliaries” and “Inherently Safer Chemistry for Accident Prevention”. Click here to view its DOZN scorecard.
应用
4-Acetamido-2,2,6,6-tetramethylpiperidine 1-oxyl radical may be employed in the catalytic system used for the regioselective oxidation of curdlan. It may be employed as model nitroxyl radical to study its reaction with thiophosgene. Reaction afforded 2,2,6,6-tetramethylpiperidine and 2,2,6,6-tetramethyl-1-hydroxypiperidine as major products.
其他说明
将醇轻度氧化成羰基的新型氧化剂
警示用语:
Warning
危险声明
危险分类
Acute Tox. 4 Oral
储存分类代码
11 - Combustible Solids
WGK
WGK 1
闪点(°F)
Not applicable
闪点(°C)
Not applicable
个人防护装备
dust mask type N95 (US), Eyeshields, Gloves
历史批次信息供参考:
分析证书(COA)
Lot/Batch Number
其他客户在看
A 1H, 13C and 15N NMR study of oxidation and reduction products of 4-acetamido-2, 2, 6, 6-tetramethylpiperidine-1-oxyl.
Schilf W, et al.
Journal of Molecular Structure, 407(1), 1-3 (1997)
Z. Ma et al.
The Journal of Organic Chemistry, 56, 6110-6110 (1991)
Reaction of Nitroxides with Sulfur Containing Compounds II [1]. Preparation of Nitroxides Bearing an Isothiocyanate Substituent in View of the Nitroxyl Group Reduction with Thiophosgene.
Zakrzewski J, et al.
Monatshefte fur Chemie / Chemical Monthly, 134(6), 843-850 (2003)
D Metodiewa et al.
Biochemistry and molecular biology international, 42(6), 1261-1270 (1997-09-26)
A novel complex, Rutoxyl [rutin/4-acetamide-1-hydroxy-2,2,6,6-tetramethylpiperidinium] was synthesized and its structure and anticancer activity were investigated. The results reported here are consistent with our idea, that the formation of such a complex of two biologically active molecules: polyphenolic flavonoid antioxidant (Rutin)
D Metodiewa et al.
Anticancer research, 19(2A), 1255-1260 (1999-06-16)
Since flavanone oximes derivatives (ethers) have been shown to modulate the growth of Yoshida Sarcoma cells in vivo and to induce apoptosis, the effects of these substances on immortalized cell lines growth were examined. Cell viability and sensitivity to investigated
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门