所有图片(2)
About This Item
经验公式(希尔记法):
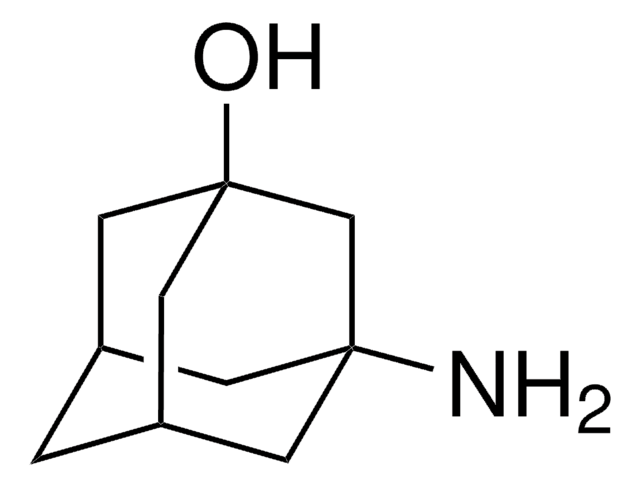
C10H17N · HCl
CAS号:
分子量:
187.71
Beilstein:
4297901
EC號碼:
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
品質等級
化驗
99%
形狀
solid
mp
>300 °C (lit.)
SMILES 字串
Cl.NC1[C@@H]2C[C@H]3C[C@@H](C2)C[C@@H]1C3
InChI
1S/C10H17N.ClH/c11-10-8-2-6-1-7(4-8)5-9(10)3-6;/h6-10H,1-5,11H2;1H/t6-,7+,8-,9+,10?;
InChI 密鑰
WLDWDRZITJEWRJ-ZDAMNCSYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
The HPLC assay of 2-adamantylamine hydrochloride after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole has been studied.
應用
2-Adamantylamine hydrochloride was used to prepare 2-adamantylamide of 2′-(carboxymethoxime)-olivomycin I.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
S V Krapivin et al.
Biulleten' eksperimental'noi biologii i meditsiny, 116(11), 515-518 (1993-11-01)
The action of the new stimulant bromantane on spectra power EEG on Fourier of sensorimotor cortex, dorsal hippocamp and lateral hypothalamus of left and right hemispheres of brain of rat in free behavior was investigated. Bromantane leads to decreases in
Alina A Okhina et al.
Journal of pharmaceutical and biomedical analysis, 180, 113039-113039 (2019-12-23)
A method of quantitative determination of camphecene, a new anti-influenza agent, in rat blood plasma based on LC-MS/MS was developed, validated and used to study the distribution of the agent between blood cells and blood plasma. The method was validated
C Clark et al.
Immunopharmacology, 21(1), 41-50 (1991-01-01)
The present in vitro investigations on amantadine (AmTd) and its isomer 2-aminoadamantane (2-NH2-Adam), and the corresponding analogs, 1-nitroadamantane (1-NO2-Adam) and 2-nitroadamantane (2-NO2-Adam), were undertaken to gain information about molecular features that might have a dominant role in inhibiting T lymphocyte
Yasuhiko Higashi et al.
Biomedical chromatography : BMC, 20(5), 423-428 (2005-09-15)
Simultaneous HPLC assay of 1-adamantanamine hydrochloride (amantadine) and its four related compounds [2-adamantanamine hydrochloride (2-ADA), 1-adamantanmethylamine (ADAMA), 1-(1-adamantyl)ethylamine hydrochloride (rimantadine) and 3,5-dimethyl-1-adamantanamine hydrochloride (memantine)] in phosphate-buffered saline (pH 7.4) after pre-column derivatization with 4-fluoro-7-nitro-2,1,3-benzoxadiazole (NBD-F) was developed. Phosphate-buffered saline samples
Anna N Tevyashova et al.
The Journal of antibiotics, 62(1), 37-41 (2009-01-10)
A novel way of chemical modification of the antibiotic olivomycin I at the 2'-keto group of the side chain of the aglycone moiety was developed. Reaction of olivomycin I with the carboxymethoxylamine hemihydrochloride gave the key intermediate, 2'-carboxymethoxime-olivomycin I, which
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持









![1-Bicyclo[1.1.1]pentylamine hydrochloride](/deepweb/assets/sigmaaldrich/product/structures/287/052/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21/640/55f4f60a-a9e0-4ea2-b1e8-5b3f6ce0ff21.png)

