S1826
Sialic Acid Aldolase from Escherichia coli K12
recombinant, expressed in E. coli BL21, ≥3.0 units/mg protein
Synonyma:
N-Acetylneuraminate lyase, N-Acetylneuraminate pyruvate-lyase (N-acetyl-D-mannosamine-forming)
About This Item
Doporučené produkty
recombinant
expressed in E. coli BL21
Quality Level
form
lyophilized powder
specific activity
≥3.0 units/mg protein
mol wt
33.4 kDa
shipped in
dry ice
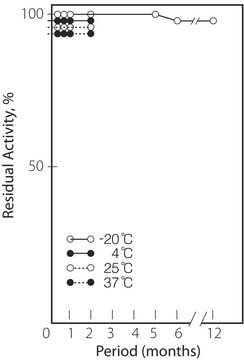
storage temp.
−20°C
General description
Application
Unit Definition
Physical form
Analysis Note
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Vyberte jednu z posledních verzí:
Osvědčení o analýze (COA)
Nevidíte správnou verzi?
Potřebujete-li konkrétní verzi, můžete vyhledat daný certifikát podle čísla dávky nebo čísla šarže.
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Zákazníci si také prohlíželi
Sortimentní položky
Understand sialic acid structure, function, signaling, and modifications. Easily find products for sialic acid research.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.