I0060000
Ifosfamide
European Pharmacopoeia (EP) Reference Standard
Synonyma:
Ifex, N,3-Bis(2-chloroethyl)tetrahydro-2H-1,3,2-oxazaphosphorin-2-amine-2-oxide
About This Item
Doporučené produkty
grade
pharmaceutical primary standard
API family
ifosfamide
manufacturer/tradename
EDQM
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
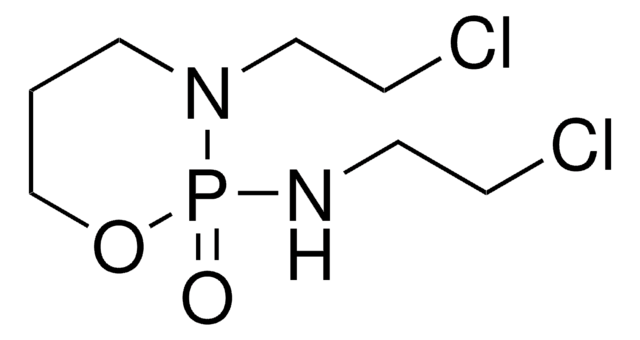
SMILES string
ClCCNP1(=O)OCCCN1CCCl
InChI
1S/C7H15Cl2N2O2P/c8-2-4-10-14(12)11(6-3-9)5-1-7-13-14/h1-7H2,(H,10,12)
InChI key
HOMGKSMUEGBAAB-UHFFFAOYSA-N
Hledáte podobné produkty? Navštivte Průvodce porovnáváním produktů
General description
Application
Biochem/physiol Actions
Packaging
Other Notes
related product
signalword
Danger
Hazard Classifications
Acute Tox. 3 Oral - Carc. 1B - Eye Irrit. 2 - Muta. 1B - Repr. 1B
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Vyberte jednu z posledních verzí:
Osvědčení o analýze (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumenty section.
Potřebujete-li pomoc, obraťte se na Zákaznická podpora
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.