913030
trYPhos™
Umicore
Synonyma:
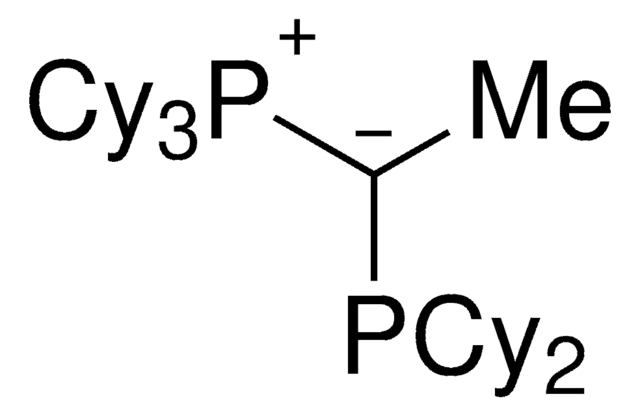
(CyYPhos)(Me)PtBu2, Tricyclohexyl(1-(di-tert-butylphosphanyl)ethylidene)phosphane
About This Item
Doporučené produkty
Quality Level
form
powder
reaction suitability
reagent type: ligand
mp
137-139 °C
functional group
phosphine
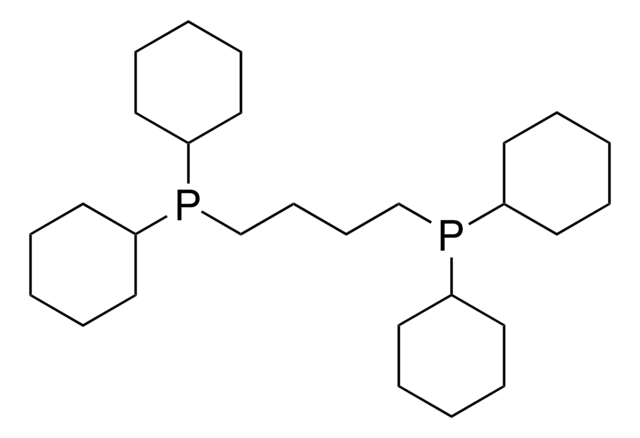
SMILES string
[P](=C(P(C(C)(C)C)C(C)(C)C)C)(C3CCCCC3)(C2CCCCC2)C1CCCCC1
InChI
1S/C28H54P2/c1-23(29(27(2,3)4)28(5,6)7)30(24-17-11-8-12-18-24,25-19-13-9-14-20-25)26-21-15-10-16-22-26/h24-26H,8-22H2,1-7H3
InChI key
TYHSTNKVOIFVOF-UHFFFAOYSA-N
General description
Application
Learn more about ylide-functionalized phosphines (YPhos)
Features and Benefits
Legal Information
This product, its manufacturing or use, is the subject of one or more issued or pending U.S. Patents (and foreign equivalents) owned or controlled by Umicore PMC. The purchase of this product from Umicore PMC through Sigma-Aldrich, its affiliates or their authorized distributors conveys to the buyer a limited, one-time, non-exclusive, non-transferable, non-assignable license. Buyer′s use of this product may infringe patents owned or controlled by third parties. It is the sole responsibility of buyer to ensure that its use of the product does not infringe the patent rights of third parties or exceed the scope of the license granted herein.
For any further information on product please refer to your local Umicore PMC contact at www.pmc.umicore.com.
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Vyberte jednu z posledních verzí:
Osvědčení o analýze (COA)
Je nám líto, ale pro tento produkt momentálně nemáme COA k dispozici online.
Potřebujete-li pomoc, obraťte se na Zákaznická podpora
Již tento produkt vlastníte?
Dokumenty související s produkty, které jste v minulosti zakoupili, byly za účelem usnadnění shromážděny ve vaší Knihovně dokumentů.
Sortimentní položky
Explore innovative palladium-catalyzed coupling reactions with ylide-substituted phosphines. Learn about their impressive capabilities, enabling milder conditions and access to aryl chlorides.
YPhos ligands enable efficient palladium-catalyzed coupling reactions under mild conditions, enhancing the synthesis of complex organic molecules.
Náš tým vědeckých pracovníků má zkušenosti ve všech oblastech výzkumu, včetně přírodních věd, materiálových věd, chemické syntézy, chromatografie, analytiky a mnoha dalších..
Obraťte se na technický servis.