A1879
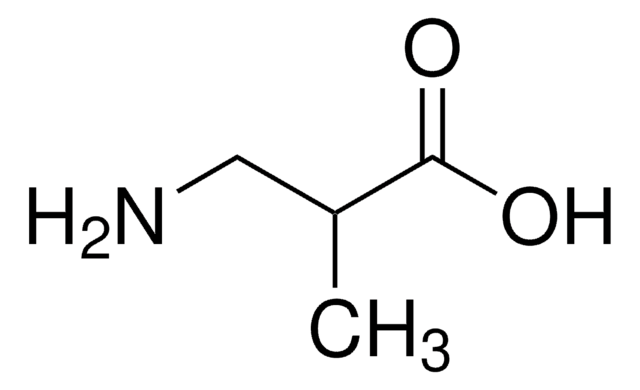
L-2-Aminobutyric acid
≥99% (titration), suitable for ligand binding assays
Sinónimos:
L-α-Aminobutyric acid
About This Item
Productos recomendados
product name
L-2-Aminobutyric acid, ≥99% (titration)
Quality Level
assay
≥99% (titration)
form
powder
technique(s)
ligand binding assay: suitable
color
white
application(s)
peptide synthesis
SMILES string
CC[C@H](N)C(O)=O
InChI
1S/C4H9NO2/c1-2-3(5)4(6)7/h3H,2,5H2,1H3,(H,6,7)/t3-/m0/s1
InChI key
QWCKQJZIFLGMSD-VKHMYHEASA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- Targeted metabolomics combined with machine learning to identify and validate new biomarkers for early SLE diagnosis and disease activity.: This study utilizes L-2-aminobutyric acid in targeted metabolomics, enhancing the predictive accuracy for Systemic Lupus Erythematosus (SLE), a crucial development in autoimmune disease diagnostics (Liang J et al., 2024).
- Alcoholamine enhanced fractionation of cellulose from lignocellulosic biomass in ionic liquids.: This research highlights the use of L-2-aminobutyric acid in the process of biomass conversion, offering insights into more efficient biofuel production techniques (Zhu Y et al., 2023).
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico