90827
Tributylphosphine
≥93.5% (Tri-N-butylphosphine, GC)
Sinónimos:
P(n-Bu)3, TBP
About This Item
Productos recomendados
vapor density
9 (vs air)
Quality Level
assay
≥93.5% (Tri-N-butylphosphine, GC)
≥97% (Tri-N-butylphospine + isomers)
form
liquid
autoignition temp.
392 °F
reaction suitability
reaction type: Acetylations
reagent type: ligand
refractive index
n20/D 1.462 (lit.)
n20/D 1.463
bp
150 °C/50 mmHg (lit.)
density
0.81 g/mL at 25 °C (lit.)
functional group
phosphine
SMILES string
CCCCP(CCCC)CCCC
InChI
1S/C12H27P/c1-4-7-10-13(11-8-5-2)12-9-6-3/h4-12H2,1-3H3
InChI key
TUQOTMZNTHZOKS-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
It may be used in the following processes:
- As reducing agent for alkyl disulfides and aromatic disulfides.
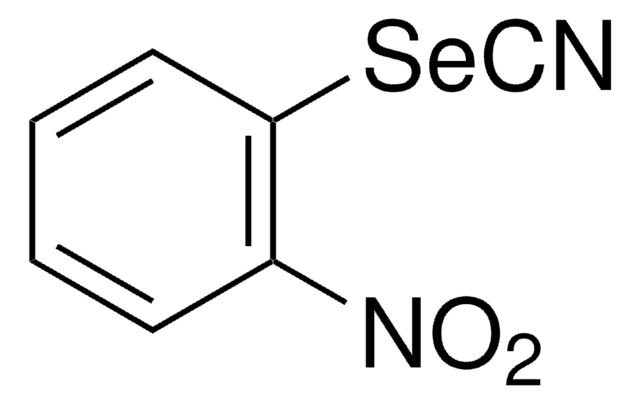
- As catalyst for the synthesis of 2-substituted 1,3-benzoselenazoles.
- As promoter for the ring opening of epoxides and aziridines with nucleophiles.
- As a reagent in the preparation of 6-substituted penicillanate esters by reduction of 6-bromo-6-substituted penicillanate esters in high diastereoselectivity.
- As a catalyst in the acylation reaction of alcohols.
- As a catalyst to prepare rotaxanes by the acylation of corresponding pseudorotaxanes using 3,5-dimethylbenzoic anhydride.
- As a catalyst to prepare vinyl thioethers by the Michael addition of ethanethiol to various alkynyl ketones.
- As a promoter in the conjugate addition of non-nucleophilic N-containing compounds with Michael acceptors.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Pyr. Liq. 1 - Skin Corr. 1A
Storage Class
4.2 - Pyrophoric and self-heating hazardous materials
wgk_germany
WGK 2
flash_point_f
242.6 °F - closed cup
flash_point_c
117 °C - closed cup
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico