276030
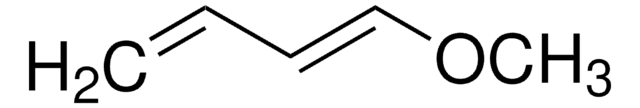
2,3-Dimethoxy-1,3-butadiene
95%
Sinónimos:
2,3-Dimethoxybuta-1,3-diene, 2,3-Dimethoxybutadiene
About This Item
Productos recomendados
assay
95%
form
liquid
refractive index
n20/D 1.459 (lit.)
bp
134-136 °C/745 mmHg (lit.)
mp
19 °C (lit.)
density
0.94 g/mL at 25 °C (lit.)
functional group
ether
storage temp.
2-8°C
SMILES string
COC(=C)C(=C)OC
InChI
1S/C6H10O2/c1-5(7-3)6(2)8-4/h1-2H2,3-4H3
InChI key
NHBDKDZHQKQPTF-UHFFFAOYSA-N
General description
Application
It may be used in the preparation of 3,4-dimethoxythiophene, an intermediate for the synthesis of 3,4-ethylenedioxythiophene (EDOT). It may also be used to form thio esters by reacting with mercaptans in the presence of cobalt carbonyl catalyst.
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico