C3743
CI 976
>98% (HPLC), solid
Synonyme(s) :
2,2-Dimethyl-N-(2,4,6-trimethoxyphenyl)dodecanamide
About This Item
Produits recommandés
Niveau de qualité
Pureté
>98% (HPLC)
Forme
solid
Solubilité
DMSO: >10 mg/mL
H2O: insoluble <2 mg/mL
Température de stockage
2-8°C
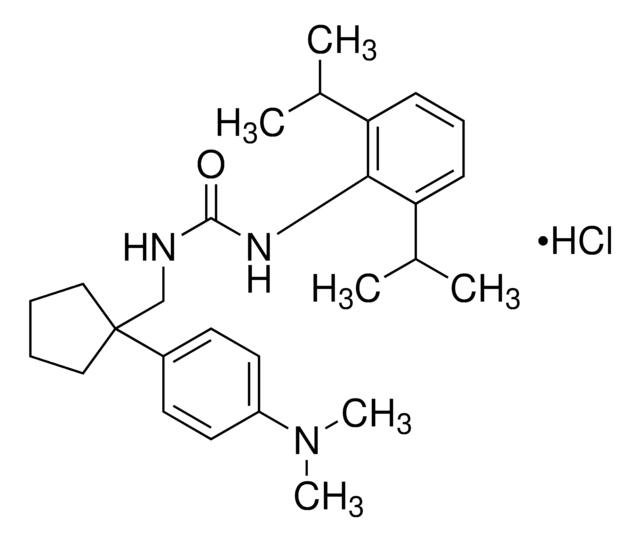
Chaîne SMILES
CCCCCCCCCCC(C)(C)C(=O)Nc1c(OC)cc(OC)cc1OC
InChI
1S/C23H39NO4/c1-7-8-9-10-11-12-13-14-15-23(2,3)22(25)24-21-19(27-5)16-18(26-4)17-20(21)28-6/h16-17H,7-15H2,1-6H3,(H,24,25)
Clé InChI
WAFNZAURAWBNDZ-UHFFFAOYSA-N
Application
- to analyze its anti-hepatitis C virus (HCV) activity in Huh7.5.1 cells
- to treat Neuro-2a cells to test its effect on plasma membrane integrated density of α4-SEPβ2 or α6-SEPβ2β3 nicotinic acetylcholine receptors (nAChRs)
- to study its effects on the anti-angiogenic activity of pyripyropenes in human umbilical vein endothelial cells
Actions biochimiques/physiologiques
Mentions de danger
Conseils de prudence
Classification des risques
Aquatic Chronic 4
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique