Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 1 g | Check Cart for Availability | CLP 411,000 |
About This Item
form
powder
Quality Level
mp
208-210 °C (lit.)
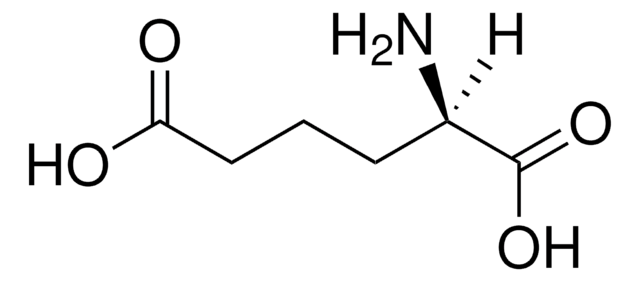
SMILES string
N[C@H](CCCC(O)=O)C(O)=O
InChI
1S/C6H11NO4/c7-4(6(10)11)2-1-3-5(8)9/h4H,1-3,7H2,(H,8,9)(H,10,11)/t4-/m1/s1
InChI key
OYIFNHCXNCRBQI-SCSAIBSYSA-N
Gene Information
rat ... Grin2a(24409), Grm1(24414), Grm2(24415), Grm6(24419)
Application
1 of 1
This Item | |||
|---|---|---|---|
| form powder | form powder | form powder | form powder |
| Quality Level 200 | Quality Level 200 | Quality Level 100 | Quality Level 100 |
| Gene Information rat ... Grin2a(24409), Grm1(24414), Grm2(24415), Grm6(24419) | Gene Information human ... GLUL(2752), LGSN(51557) | Gene Information rat ... Slc1a3(29483) | Gene Information - |
| mp 208-210 °C (lit.) | mp 203-205 °C (dec.) (lit.) | mp >300 °C (lit.) | mp >295 °C (subl.) (lit.) |
Still not finding the right product?
Explore all of our products under D-2-Aminoadipic acid
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| A7400-1G | 04061826688434 |