Fortfahren mit
Größe auswählen
| Ihnen/SKU | Verfügbarkeit | Preis |
|---|---|---|
25 g | Warenkorb auf Verfügbarkeit prüfen | € 43,20 |
100 g | Warenkorb auf Verfügbarkeit prüfen | € 144,00 € 122,40 |
250 g | Warenkorb auf Verfügbarkeit prüfen | € 283,00 € 240,55 |
1 kg | Warenkorb auf Verfügbarkeit prüfen | € 886,00 |
Über diesen Artikel
€ 43,20
Produktname
2-Thiouracil, ≥99%
biological source
synthetic
Quality Segment
assay
≥99%
form
powder
mp
>300 °C (lit.)
solubility
1 M NaOH: 5 mg/mL, clear to very slightly hazy, 1 M NaOH: 50 mg/mL, colorless to faintly yellow
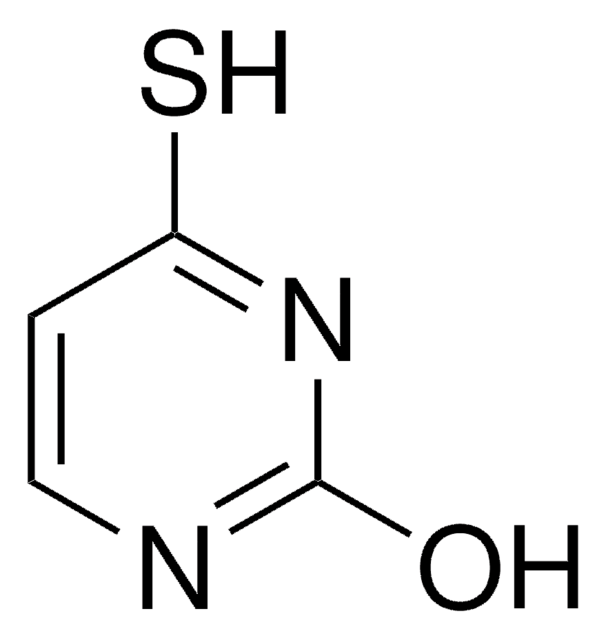
SMILES string
O=C1NC(=S)NC=C1
InChI
1S/C4H4N2OS/c7-3-1-2-5-4(8)6-3/h1-2H,(H2,5,6,7,8)
InChI key
ZEMGGZBWXRYJHK-UHFFFAOYSA-N
General description
Application
- in the synthesis and characterization of silver colloid and film substrates and their applications in surface-enhanced Raman scattering (SERS)
- to study its electro-oxidation and determination at titanium dioxide (TiO2) nanoparticles-modified gold electrode
- to study the effect of methylation on the deactivation mechanism or the triplet-state dynamics of 2-thiouracil using time-resolved photoelectron spectroscopy
Biochem/physiol Actions
1 of 1
Dieser Artikel | |||
|---|---|---|---|
| biological source synthetic | biological source synthetic (organic) | biological source - | biological source synthetic (organic) |
| Quality Level 200 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| assay ≥99% | assay ≥98% (TLC) | assay 97% | assay ~98% |
| form powder | form powder | form powder | form lyophilized powder |
| solubility 1 M NaOH: 5 mg/mL, clear to very slightly hazy, 1 M NaOH: 50 mg/mL, colorless to faintly yellow | solubility 1 M NaOH: 50 mg/mL, clear, colorless to faintly yellow | solubility 1 M NaOH: soluble 50 mg/mL | solubility 1 M NaOH: 50 mg/mL, clear to slightly hazy |
| mp >300 °C (lit.) | mp - | mp 295 °C (dec.) (lit.) | mp ≥300 °C (lit.) |
Still not finding the right product?
Explore all of our products under 2-Thiouracil
signalword
Warning
hcodes
Hazard Classifications
Carc. 2
Lagerklasse
11 - Combustible Solids
wgk
WGK 3
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.