121231
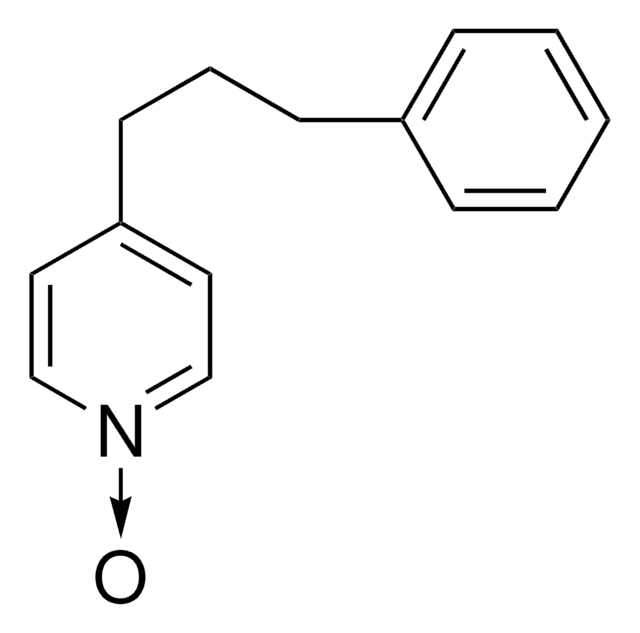
4-(3-Phenylpropyl)pyridin
97%
About This Item
Empfohlene Produkte
Assay
97%
Brechungsindex
n20/D 1.563 (lit.)
bp
322 °C (lit.)
Löslichkeit
H2O: insoluble
Dichte
1.03 g/mL at 25 °C (lit.)
SMILES String
C(Cc1ccccc1)Cc2ccncc2
InChI
1S/C14H15N/c1-2-5-13(6-3-1)7-4-8-14-9-11-15-12-10-14/h1-3,5-6,9-12H,4,7-8H2
InChIKey
AQIIVEISJBBUCR-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
Anwendung
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
10 - Combustible liquids
WGK
WGK 3
Flammpunkt (°F)
235.4 °F - closed cup
Flammpunkt (°C)
113 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.


![4-[6-(4-pyridinyl)hexyl]pyridine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/969/304/69102a98-4497-4db9-b5d7-792b9c6fb5e7/640/69102a98-4497-4db9-b5d7-792b9c6fb5e7.png)