L6545
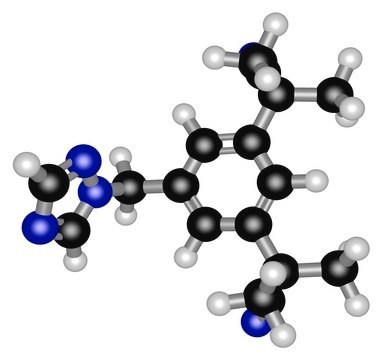
Letrozole
≥98% (HPLC), powder, non-steroidal aromatase inhibitor
Sinónimos:
4,4′-(1H-1,2,4-Triazol-1-ylmethylene)bisbenzonitrile
Seleccione un Tamaño
Seleccione un Tamaño
About This Item
Productos recomendados
Nombre del producto
Letrozole, ≥98% (HPLC)
Ensayo
≥98% (HPLC)
Formulario
powder
color
white to off-white
solubilidad
DMSO: >50 mg/mL
emisor
Novartis
temp. de almacenamiento
2-8°C
cadena SMILES
N#CC(C=C1)=CC=C1C(N2C=NC=N2)C3=CC=C(C#N)C=C3
InChI
1S/C17H11N5/c18-9-13-1-5-15(6-2-13)17(22-12-20-11-21-22)16-7-3-14(10-19)4-8-16/h1-8,11-12,17H
Clave InChI
HPJKCIUCZWXJDR-UHFFFAOYSA-N
Información sobre el gen
human ... CYP19A1(1588)
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
- in organoid growth assay to determine its inhibitory capacity(48)
- to investigate steroid receptor coactivator-1 (SRC-1) mediated endogenous estrogen regulation of hippocampal PSD-95(49)
- to determine its effects on tumor-induced hyperalgesia(50)
- for hormonal manipulation in rats(51)
- to study its effects on lipocalin-2 (Lcn2)(52)
- to determine its effects on mechanical hyperalgesia and aromatase expression(53)
Acciones bioquímicas o fisiológicas
Características y beneficios
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Repr. 2 - STOT RE 2
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico