All Photos(1)
About This Item
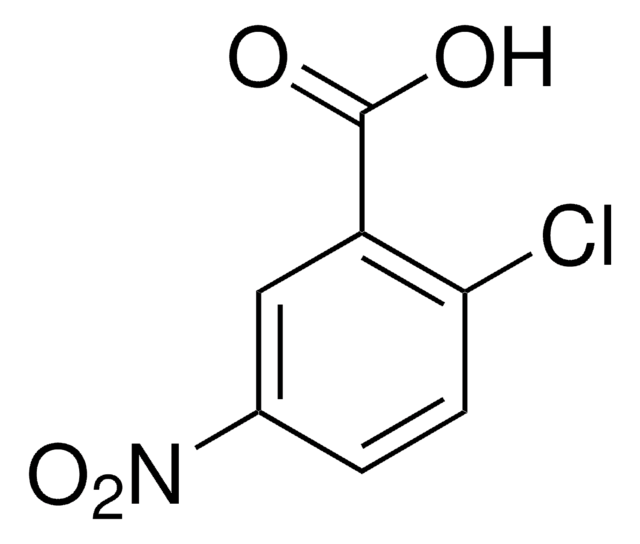
Linear Formula:
ClC6H3(NO2)CO2H
CAS Number:
Molecular Weight:
201.56
Beilstein:
1877474
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
powder
mp
165-168 °C (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)c1cc(ccc1Cl)[N+]([O-])=O
InChI
1S/C7H4ClNO4/c8-6-2-1-4(9(12)13)3-5(6)7(10)11/h1-3H,(H,10,11)
InChI key
QUEKGYQTRJVEQC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2-Chloro-5-nitrobenzoic acid undergoes microwave-assisted, regioselective amination reaction with aliphatic and aromatic amines to yield N-substituted 5-nitroanthranilic acid derivatives. It acts as ligand and forms a red luminescent one dimensional coordination polymer with Eu(III).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
2-Chloro-5-nitrobenzoato complexes of Eu (III) and Tb (III)-A 1D coordination polymer and enhanced solution luminescence.
Viswanathan S and Bettencourt-Dias A.
Inorganic Chemistry Communications, 9(5), 444-448 (2006)
Younis Baqi et al.
The Journal of organic chemistry, 72(15), 5908-5911 (2007-06-26)
The synthesis of N-substituted 5-nitroanthranilic acid derivatives 3a-w was achieved by a new, mild, microwave-assisted, regioselective amination reaction of 5-nitro-2-chlorobenzoic acid (1a) with a diverse range of aliphatic and aromatic amines 2a-w without added solvent or catalyst. Up to >99%
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service