All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10H14O2
CAS Number:
Molecular Weight:
166.22
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
98%
form
solid
mp
>300 °C (lit.)
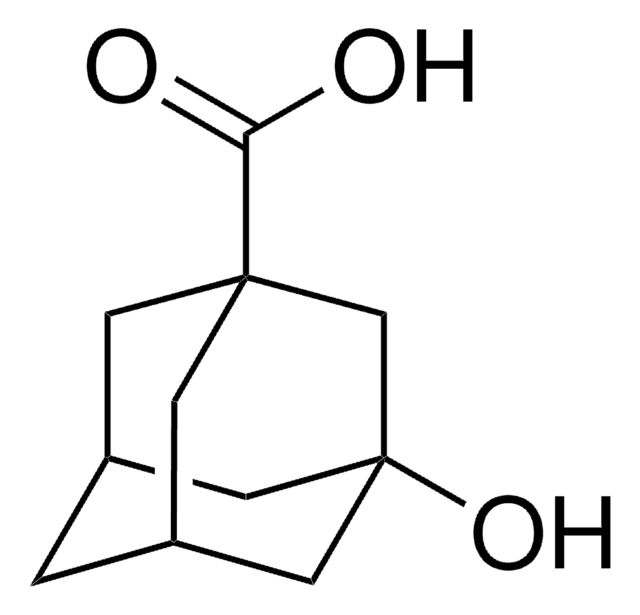
SMILES string
O[C@]12C[C@@H]3C[C@H](C1)C(=O)[C@@H](C3)C2
InChI
1S/C10H14O2/c11-9-7-1-6-2-8(9)5-10(12,3-6)4-7/h6-8,12H,1-5H2/t6-,7-,8+,10-
InChI key
TZBDEVBNMSLVKT-XYYXLIQBSA-N
General description
5-Hydroxy-2-adamantanone is a disubstituted derivative of adamantane. The biocatalyzed synthesis of 5-hydroxy-2-adamantanone from 2-adamantanone has been investigated.
Application
5-Hydroxy-2-adamantanone may be used in the following studies:
- As a model compound to investigate the application of lanthanide NMR shift reagents for the analysis of disubstituted derivative of adamantane.
- As a starting material for the synthesis of E-2-amino-5-hydroxyadamantane.
- As a starting material for the synthesis of 4-(triphenylsilyloxy)adamantan-1-ol.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
[The immunomodulator kemantan in the treatment of patients with exacerbated chronic obstructive bronchitis].
E M Rekalova
Likars'ka sprava, (4)(4), 73-76 (1992-04-01)
S S Boĭko et al.
Farmakologiia i toksikologiia, 54(1), 57-59 (1991-01-01)
The pharmacokinetics of a new Soviet-made immunostimulant kemantane, a derivative of adamantine, was studied by gas-liquid chromatography in patients with bronchial pathology. It was found that in the blood of the patients kemantane was not practically detected due to a
An expeditious preparation of E-2-amino-5-hydroxyadamantane and its Z-isomer.
Jaroskova L, et al.
Tetrahedron Letters, 47(46), 8063-8067 (2006)
Marta L Lage et al.
Tetrahedron, 69(27-28), 5609-5613 (2013-09-03)
A chemoselective method for the hydrosilylation of ketones has been developed, using the combination of triphenylsilane and a catalyst prepared from Ni(COD)
Application of shift reagents in the study of disubstituted derivatives of adamantane by NMR spectroscopy.
Vodicka L, et al.
Collection of Czechoslovak Chemical Communications, 40(1), 293-299 (1975)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service