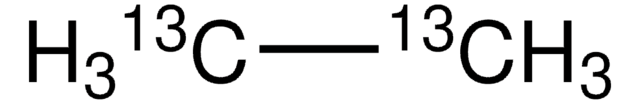About This Item
Recommended Products
vapor density
1.05 (vs air)
vapor pressure
37.95 atm ( 21.1 °C)
assay
99.99%
form
gas
autoignition temp.
881 °F
expl. lim.
13 %
bp
−88 °C (lit.)
mp
−172 °C (lit.)
density
0.362 g/mL at 20 °C (lit.)
SMILES string
CC
InChI
1S/C2H6/c1-2/h1-2H3
InChI key
OTMSDBZUPAUEDD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- High-pressure oxidation of ethane: The paper discusses the oxidation properties of ethane under high pressure, providing insights crucial for developing combustion models and understanding ethane′s behavior in various industrial processes (H Hashemi, JG Jacobsen, CT Rasmussen, 2017).
- Progress and prospects in catalytic ethane aromatization: This review highlights the advancements in converting ethane to more valuable aromatic hydrocarbons, showcasing the potential of ethane as a petrochemical feedstock (Y Xiang, H Wang, J Cheng, J Matsubu, 2018).
Packaging
Compatible with the following:
- Aldrich® lecture-bottle station systems
- Aldrich® lecture-bottle gas regulators
Other Notes
Legal Information
also commonly purchased with this product
control valve
hose barb
purge valve
recommended
signalword
Danger
hcodes
Hazard Classifications
Flam. Gas 1A - Press. Gas Liquefied gas
Storage Class
2A - Gases
wgk_germany
nwg
flash_point_f
-211.0 °F - closed cup
flash_point_c
-135 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, multi-purpose combination respirator cartridge (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Separation of Methane; Acetylene; Carbon monoxide; Water; Nitrogen; Carbon dioxide; Ethane; Ethylene
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service