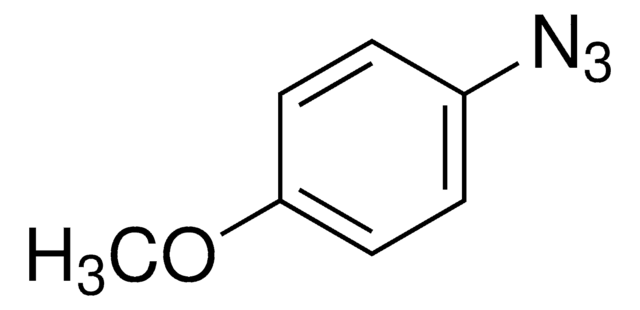
727490
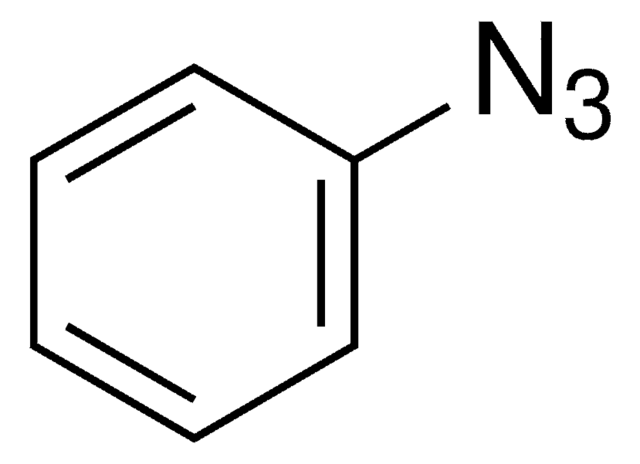
Azidobenzene solution
~0.5 M in tert-butyl methyl ether
Synonym(s):
Phenyl azide solution
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H5N3
CAS Number:
Molecular Weight:
119.12
Beilstein:
742248
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥95.0% (HPLC)
Quality Level
form
liquid
concentration
~0.5 M in tert-butyl methyl ether
impurities
≤2.0% water
functional group
azide
storage temp.
−20°C
SMILES string
[N-]=[N+]=Nc1ccccc1
InChI
1S/C6H5N3/c7-9-8-6-4-2-1-3-5-6/h1-5H
InChI key
CTRLRINCMYICJO-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 2 - Skin Irrit. 2 - STOT RE 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
39.2 °F
Flash Point(C)
4 °C
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
D Wild
Chemico-biological interactions, 82(1), 123-132 (1992-03-01)
Photolysis of arylazides produces short-lived reactive species, very likely arylnitrenium ions which bind to nucleotides and DNA and produce mutations in Salmonella. The present report shows that arylazides can be photo-activated in mammalian (V79 Chinese hamster) cells and that sister
Andrew L Hook et al.
Biomacromolecules, 10(3), 573-579 (2009-01-23)
The fabrication and characterization of chemical patterns using a technique that can be readily integrated with methods currently used for the formation of microarrays is presented. A high density poly(ethylene glycol) coating was deposited on glass slides as a background
David Evrard et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 14(30), 9286-9291 (2008-09-10)
The electrochemical reduction of phenylazide or phenylacetylene diazonium salts leads to the grafting of azido or ethynyl groups onto the surface of carbon electrodes. In the presence of copper(I) catalyst, these azide- or alkyne-modified surfaces react efficiently and rapidly with
Manabu Mizutani et al.
Biomacromolecules, 3(4), 668-675 (2002-07-09)
Photoreactive phenylazide-end-capped liquid copolymers were prepared by ring-opening copolymerization of epsilon-caprolactone (CL) and trimethylene carbonate (TMC) at an equimolar monomer feed ratio in the presence of a polyol, namely, a low-molecular-weight alcohol (di-, tri-, and tetraol) or poly(ethylene glycol) (PEG)
Mary S Rizk et al.
Biochemistry, 45(2), 543-551 (2006-01-13)
The reactive 1,2-didehydroazepine (cyclic ketenimine) intermediates produced upon photolysis of phenyl azide, 3-hydroxyphenyl azide, 3-methoxyphenyl azide, and 3-nitrophenyl azide in water and in HEPES buffer were studied by laser flash photolysis techniques with UV-vis detection of the transient intermediates. The
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service