19670
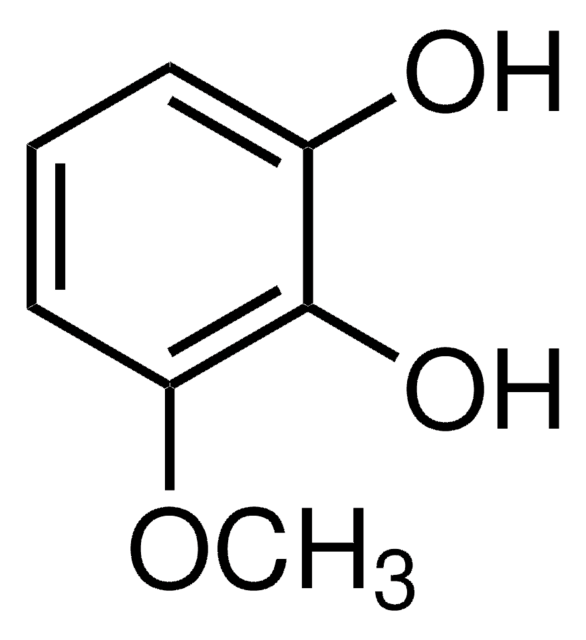
4-tert-Butylcatechol
≥97.0% (HPLC)
Sinónimos:
4-(1,1-Dimethylethyl)benzene-1,2-diol, 4-tert-Butylcatechin, 4-tert-Butylpyrocatechol, 4-TBC, p-tert-Butylcatechol
About This Item
Productos recomendados
Quality Level
assay
≥97.0% (HPLC)
bp
285 °C (lit.)
mp
52-55 °C (lit.)
53-58 °C
solubility
methanol: soluble 1 g/10 mL, clear, colorless to slightly yellow
SMILES string
CC(C)(C)c1ccc(O)c(O)c1
InChI
1S/C10H14O2/c1-10(2,3)7-4-5-8(11)9(12)6-7/h4-6,11-12H,1-3H3
InChI key
XESZUVZBAMCAEJ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- Electrochemical Oxidation of Catechol and 4-tert-Butylcatechol: This study explores the oxidation reactions of catechol and 4-tert-butylcatechol, detailing the electrochemical pathways and potential applications in synthetic chemistry (E Tammari, M Heravi, D Nematollahi, academia.edu).
- Selective Oxidation of 4-tert-butylphenol by Hydrogen Peroxide: Discusses the catalytic performance of titanosilicates in the selective oxidation to 4-tert-butylcatechol, emphasizing improvements in selectivity and yield (RR Talipova et al., Applied Petrochemical Research, 2016 - Springer).
- Barium 5-(tert-butyl)-2,3-dihydroxybenzenesulfonate: This research presents the sulfonation process of 4-tert-butylcatechol, contributing to the development of new derivatives for advanced materials (LG Rubicheva, DA Lukyanov - Molbank, 2022 - mdpi.com).
- UV-AOPs for Efficient Continuous Flow Removal of 4-tert-butylphenol: Compares various advanced oxidation processes for removing contaminants like 4-tert-butylcatechol from water, highlighting the environmental applications (S Mergenbayeva, SG Poulopoulos - Processes, 2021 - mdpi.com).
- The Study on the Degradation of 4-tert-butylphenol by Hydroxyl Radical: This article investigates the degradation pathways of 4-tert-butylcatechol, providing insights into the environmental impact and degradation mechanisms (YL Wu et al., China Environmental Science, 2016 - cabdirect.org).
signalword
Danger
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
235.4 °F - closed cup
flash_point_c
113 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico