N32601
Norcamphor
98%
Synonym(s):
2-Norbornanone, Bicyclo[2.2.1]heptan-2-one
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C7H10O
CAS Number:
Molecular Weight:
110.15
Beilstein/REAXYS Number:
1209657
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
98%
form
crystals
bp
168-172 °C (lit.)
mp
93-96 °C (lit.)
SMILES string
O=C1C[C@@H]2CC[C@H]1C2
InChI
1S/C7H10O/c8-7-4-5-1-2-6(7)3-5/h5-6H,1-4H2/t5-,6+/m1/s1
InChI key
KPMKEVXVVHNIEY-RITPCOANSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Sarasij Kumar Upadhyay et al.
The Journal of organic chemistry, 76(5), 1355-1360 (2011-01-22)
The endo/exo product ratio in the reactions of SmI(2) with norcamphor in the presence of various proton donors was determined. The effect of MeOH, EtOH, trifluoroethanol (TFE), ethylene glycol (EG), and water was investigated at various concentrations of these proton
W M Atkins et al.
Biochemistry, 27(5), 1610-1616 (1988-03-08)
The kinetics of NADH consumption, oxygen uptake, and hydrogen peroxide production have been studied for norcamphor metabolism by cytochrome P-450cam. The kinetic deuterium isotope effects on these processes, with specifically deuteriated norcamphor, are 0.77, 1.22, and 1.16, respectively. Steady-state UV-visible
Lars Goerigk et al.
The journal of physical chemistry. A, 113(4), 767-776 (2008-12-24)
Time-dependent double-hybrid density functional theory is applied to the calculation of the electronic circular dichroism (CD) spectra of molecules. The TD-B2PLYP method is based on vertical excitation energies obtained from its hybrid-GGA part B2LYP in a conventional TD-DFT linear response
M B Bass et al.
Proteins, 13(1), 26-37 (1992-05-01)
While cytochrome P-450cam catalyzes the hydroxylation of camphor to 5-exo-hydroxycamphor with 100% stereospecificity, norcamphor is hydroxylated by this enzyme yielding 45% 5-exo-, 47% 6-exo-, and 8% 3-exo-hydroxynorcamphor (Atkins, W.M., Sligar, S.G., J. Am. Chem. Soc. 109:3754-3760, 1987). The present study
P J Loida et al.
The Journal of biological chemistry, 270(10), 5326-5330 (1995-03-10)
The stereoselectivity of cytochrome P450cam hydroxylation has been investigated with the enantiomerically pure substrate analog norcamphor. (1R)- and (1S)-norcamphor (> 92 enantiomeric excess) were characterized in the hydroxylation reaction with cytochrome P450cam with respect to the product profile, steady state
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service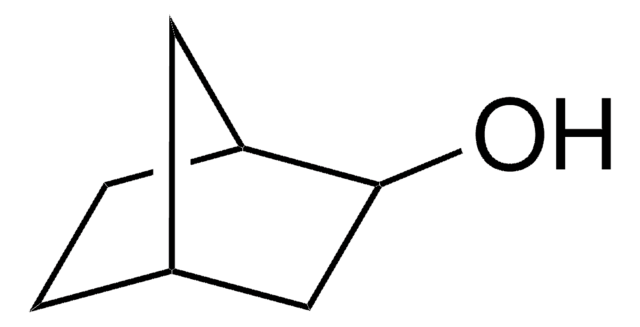





![Bicyclo[3.3.1]nonan-9-one ≥98%](/deepweb/assets/sigmaaldrich/product/structures/270/852/60661ded-13fb-4fc7-af36-a381880070a5/640/60661ded-13fb-4fc7-af36-a381880070a5.png)

![Bicyclo[2.2.1]hept-2-ene 99%](/deepweb/assets/sigmaaldrich/product/structures/270/492/95fd4909-6108-4858-8c94-609b54387149/640/95fd4909-6108-4858-8c94-609b54387149.png)
