All Photos(1)
About This Item
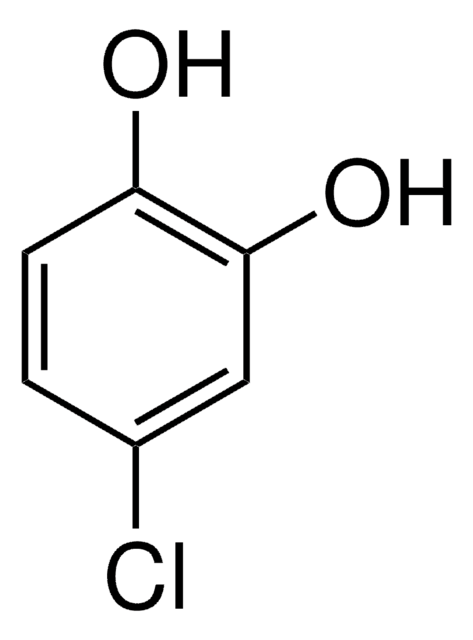
Linear Formula:
Cl2C6H2(OH)2
CAS Number:
Molecular Weight:
179.00
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
assay
98%
mp
168-171 °C (lit.)
SMILES string
Oc1cc(Cl)c(O)cc1Cl
InChI
1S/C6H4Cl2O2/c7-3-1-5(9)4(8)2-6(3)10/h1-2,9-10H
InChI key
AYNPIRVEWMUJDE-UHFFFAOYSA-N
General description
2,5-Dichlorohydroquinone (2,5-DCHQ) is a hydroquinone derivative that can be synthesized by reducing 2,5-dichloroquinone using sodium dithionite (Na2S2O4). The kinetics of the reaction of 2,5-DCHQ and N-phenyl-1,4-benzoquinonemonoimine has been studied. The transformation of 2,5-DCHQ to 2-chloromaleylacetate using PcpA (Pcp = pentachlorophenol) protein, isolated from Escherichia coli has been investigated. It is reported to be the degradation product of 2,4,5-trichlorophenoxyacetic acid and γ-hexachlorocyclohexane.
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
K Miyauchi et al.
Journal of bacteriology, 180(6), 1354-1359 (1998-03-27)
Sphingomonas (formerly Pseudomonas) paucimobilis UT26 utilizes gamma-hexachlorocyclohexane (gamma-HCH), a halogenated organic insecticide, as a sole carbon and energy source. In a previous study, we showed that gamma-HCH is degraded to 2,5-dichlorohydroquinone (2,5-DCHQ) (Y. Nagata, R. Ohtomo, K. Miyauchi, M. Fukuda
S Oikawa et al.
Carcinogenesis, 17(12), 2733-2739 (1996-12-01)
p-Dichlorobenzene (p-DCB) has been reported to be carcinogenic for rodents, although it does not seem to be mutagenic in bacterial test systems. In this study, the mechanism of DNA damage by metabolites of p-DCB in the presence of metals was
C Klos et al.
Xenobiotica; the fate of foreign compounds in biological systems, 24(10), 965-976 (1994-10-01)
1. The metabolism of 1,4-dichlorobenzene has been studied in the male and female Fisher 344 rat over 72 h after oral administration of 14C-1,4-dichlorobenzene (900 mg = 96.8 microCi/kg). No covalent binding of radioactivity could be detected in samples of
Y Nagata et al.
Journal of bacteriology, 176(11), 3117-3125 (1994-06-01)
In Pseudomonas paucimobilis UT26, gamma-hexachlorocyclohexane (gamma-HCH) is converted to 2,5-dichloro-2,5-cyclohexadiene-1,4-diol (2,5-DDOL), which is then metabolized to 2,5-dichlorohydroquinone. Here, we isolated from the genomic library of UT26 two genes which expressed 2,5-DDOL dehydrogenase activity when they were transformed into P. putida
Y Ohtsubo et al.
FEBS letters, 459(3), 395-398 (1999-10-20)
The pentachlorophenol (PCP) mineralizing bacterium Sphingomonas chlorophenolica ATCC39723 degrades PCP via 2,6-dichlorohydroquinone (2,6-DCHQ). The pathway converting PCP to 2,6-DCHQ has been established previously; however, the pathway beyond 2,6-DCHQ is not clear, although it has been suggested that a PcpA plays
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service