All Photos(1)
About This Item
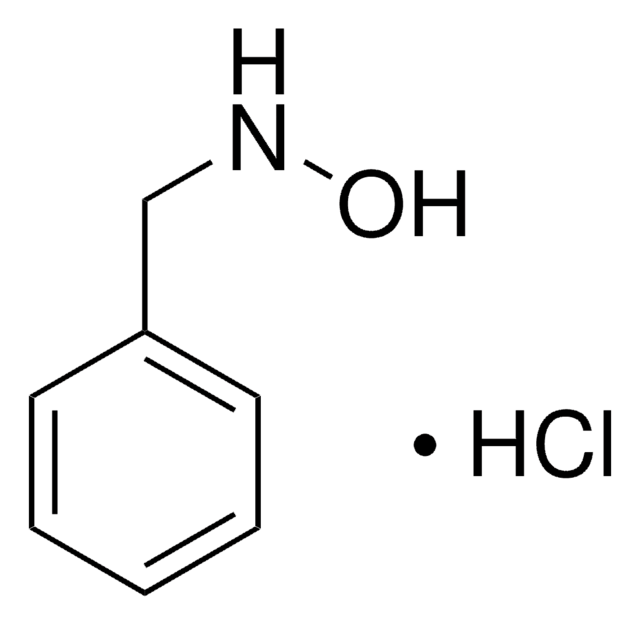
Linear Formula:
C6H5CH2NHOH · HCl
CAS Number:
Molecular Weight:
159.61
Beilstein/REAXYS Number:
507948
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
assay:
97%
Recommended Products
assay
97%
mp
108-110 °C (lit.)
functional group
amine
phenyl
SMILES string
Cl.ONCc1ccccc1
InChI
1S/C7H9NO.ClH/c9-8-6-7-4-2-1-3-5-7;/h1-5,8-9H,6H2;1H
InChI key
YSNXOQGDHGUKCZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
N-Benzylhydroxylamine hydrochloride is a N-substituted-hydroxylamine. It participates in the ring opening of (2S,3R)-1,2-epoxy-4-penten-3-ol. Two-step synthesis of N-benzylhydroxylamine starting from dibenzylamine is reported.
Application
N-Benzylhydroxylamine hydrochloride may be used in the preparation of a precursor to hitherto unknown aminocyclopentitol derivative, hydroxy functionalised 4-exo-ethoxycarbonyl-3-oxa-2-azabicyclo[3.3.0]octane system.
Analysis Note
May contain traces of methyl orange, which can cause the color to take on a faint red hue.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A Large-Scale Low-Cost Preparation of N-Benzylhydroxylamine Hydrochloride.
Nguyen TB, et al.
Synthesis, 18, 3174-3176 (2009)
[1, 3]-Dipolar intramolecular nitrone olefin cycloaddition reaction of a sugar-derived a, ?-unsaturated ester: a new diastereo-and regioselective synthesis of an aminocyclopentitol.
Jachak SM, et al.
Tetrahedron Letters, 42(29), 4925-4928 (2001)
The Synthesis of Chiral Polyhydroxylated Pyrrolidines Using a Reverse-Cope Cyclisation.
O'Neil IA, et al.
Synlett, 10, 1408-1410 (2000)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service