All Photos(2)
About This Item
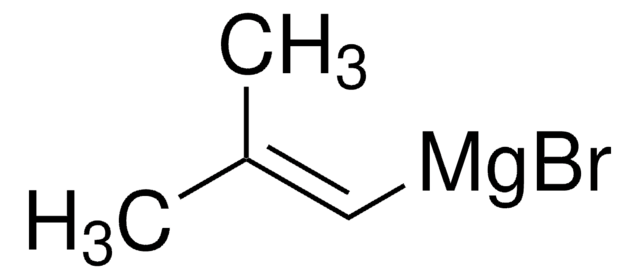
Linear Formula:
(CH3)2C=C(Br)CH3
CAS Number:
Molecular Weight:
149.03
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
96%
form
liquid
contains
copper pellets as stabilizer
refractive index
n20/D 1.474 (lit.)
bp
40 °C/75 mmHg (lit.)
density
1.284 g/mL at 25 °C (lit.)
SMILES string
C\C(C)=C(\C)Br
InChI
1S/C5H9Br/c1-4(2)5(3)6/h1-3H3
InChI key
DBELOSOZLGEZBM-UHFFFAOYSA-N
General description
2-Bromo-3-methyl-2-butene is a vinylic bromide compound. Palladium/di-1-adamantyl-n-butylphosphine-catalyzed reductive carbonylation of 2-bromo-3-methyl-2-butene has been reported. Cross-coupling reaction of 2-bromo-3-methyl-2-butene with potassium 6-(benzoyloxy)hexyltrifluoroborate and 3-(benzoyloxy)propyltrifluoroborate has been investigated.
Application
2-Bromo-3-methyl-2-butene may be used in the preparation of:
- 2,3,4,5-tetramethyl-2,4-hexadiene
- 2-iodo-3-methyl-2-butene
- diastereomers of 2-amino-3-hydroxy-4,5-dimethylhexanoic acid
- lithium reagent, 2-lithio-3-methylbut-2-ene
- D-allo-(2R,3R,4R)-2-amino-3-hydroxy-4,5-dimethylhexanoic acid-containing peptide, pipecolidepsin A.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Oral - Flam. Liq. 3
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
77.0 °F
Flash Point(C)
25 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis of All the Diastereomers of 2-Amino-3-hydroxy-4, 5-dimethylhexanoic Acid.
Spengler J and Albericio F.
European Journal of Organic Chemistry, 1, 44-47 (2014)
Marta Pelay-Gimeno et al.
Nature communications, 4, 2352-2352 (2013-08-31)
Pipecolidepsin A is a head-to-side-chain cyclodepsipeptide isolated from the marine sponge Homophymia lamellosa. This compound shows relevant cytotoxic activity in three human tumour cell lines and has unique structural features, with an abundance of non-proteinogenic residues, including several intriguing amino
Synthesis and Molecular Structure of 2, 3, 4, 5-Tetramethyl-2, 4-hexadiene.
Br M, et al.
Acta Chemica Scandinavica. Series B, 31, 387-390 (1977)
Gary A Molander et al.
Tetrahedron, 63(3), 768-775 (2008-01-16)
The Suzuki-Miyaura-type cross-coupling reaction of potassium alkyltrifluoroborates with various alkenyl bromides in the presence of 10 mol % of PdCl(2)(dppf)·CH(2)Cl(2) and 3.0 equiv of Cs(2)CO(3) in aqueous toluene at 80 °C provided the desired compounds in 49-95% yields. A variety
Palladium/di-1-adamantyl-n-butylphosphine-catalyzed reductive carbonylation of aryl and vinyl halides.
Brennfuhrer A, et al.
Tetrahedron, 63(27), 6252-6258 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service