345830
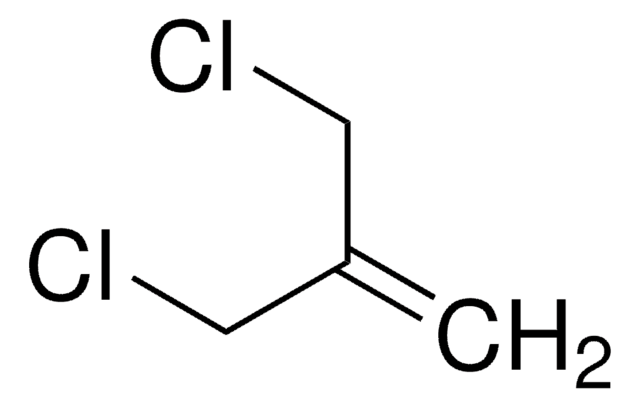
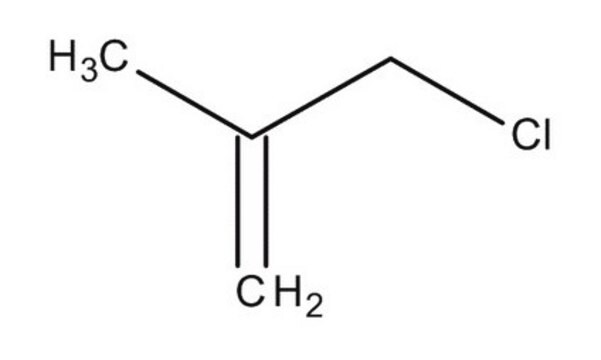
3-Bromo-2-methylpropene
97%
Synonym(s):
Methallyl bromide, Methylallyl bromide
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
H2C=C(CH3)CH2Br
CAS Number:
Molecular Weight:
135.00
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
liquid
impurities
0.1% hydroquinone
refractive index
n20/D 1.472 (lit.)
bp
94-95 °C (lit.)
density
1.339 g/mL at 25 °C (lit.)
functional group
alkyl halide
bromo
storage temp.
2-8°C
SMILES string
CC(=C)CBr
InChI
1S/C4H7Br/c1-4(2)3-5/h1,3H2,2H3
InChI key
USEGQJLHQSTGHW-UHFFFAOYSA-N
Related Categories
General description
Rotational isomerism in 3-bromo-2-methylpropene has been studied by IR spectroscopy.
Application
3-Bromo-2-methylpropene may be used:
- in the synthesis of alkenyl imines
- in the synthesis of 2-methylpropenyl (“methallyl”) complex, Cp*Os(η3-allyl)Br2
- in a study of chiral phase transfer alkylation leading to (S)-α-alkylcysteines
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
44.6 °F - closed cup
Flash Point(C)
7 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Intramolecular "hydroiminiumation" of alkenes: application to the synthesis of conjugate acids of cyclic alkyl amino carbenes (CAACs).
Rodolphe Jazzar et al.
Angewandte Chemie (International ed. in English), 46(16), 2899-2902 (2007-03-14)
Synthesis of Hydride and Alkyl Compounds Containing the Cp* Os (η3-allyl) Fragment. Crystal Structures of Cp* Os (η3-C8H13) Br2 and [Cp* Os (η3-C4H7) Me (H2O)][BF4].
Mui HD, et al.
Organometallics, 18(17), 3264-3272 (1999)
Rotational isomerism in 3-bromo, 2-methylpropene studied by ir spectroscopy.
Diallo AO.
Spectrochimica Acta. Part A, Molecular and Biomolecular Spectroscopy, 36(9), 799-803 (1980)
Taek-Soo Kim et al.
The Journal of organic chemistry, 71(21), 8276-8278 (2006-10-10)
We reported efficient enantioselective synthetic methodologies for (R)-alpha-alkylcysteines and (S)-alpha-alkylcysteines. The phase-transfer catalytic alkylation of 2-phenyl-2-thiazoline-4-carboxylic acid tert-butyl ester and 2-o-biphenyl-2-thiazoline-4-carboxylic acid tert-butyl ester, in the presence of chiral catalysts (1 or 2), gave the corresponding alkylated products, which could
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service