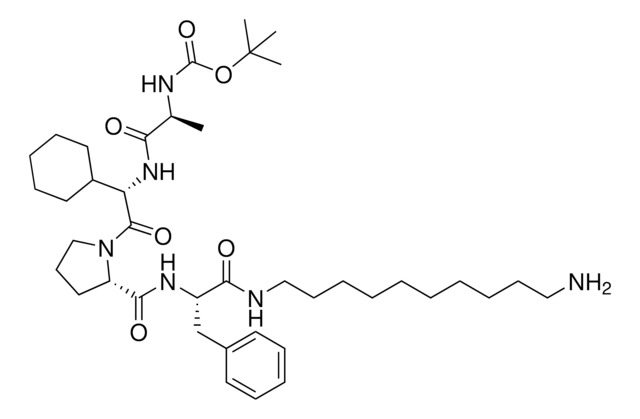
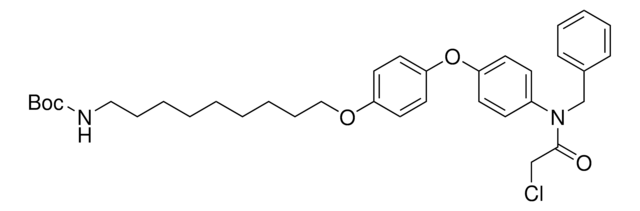
919489
CCW16-C6-PEG3-butyl-BocNH
同義詞:
tert-butyl (6-(2-(2-((6-(4-(4-(N-Benzyl-2-chloroacetamido)phenoxy)phenoxy)hexyl)oxy)ethoxy)ethoxy)hexyl)carbamate, Crosslinker-E3 Ligase ligand conjugate, Protein degrader building block for PROTAC® research, RNF4-targeting building block, Template for synthesis of targeted protein degrader
About This Item
推薦產品
ligand
CCW16
品質等級
形狀
viscous liquid
反應適用性
reactivity: carboxyl reactive
reagent type: ligand-linker conjugate
官能基
amine
儲存溫度
2-8°C
SMILES 字串
O=C(CCl)N(CC1=CC=CC=C1)C2=CC=C(C=C2)OC3=CC=C(OCCCCCCOCCOCCOCCCCCCNC(OC(C)(C)C)=O)C=C3
InChI
1S/C42H59ClN2O8/c1-42(2,3)53-41(47)44-25-11-4-5-12-26-48-29-31-50-32-30-49-27-13-6-7-14-28-51-37-21-23-39(24-22-37)52-38-19-17-36(18-20-38)45(40(46)33-43)34-35-15-9-8-10-16-35/h8-10,15-24H,4-7,11-14,25-34H2,1-3H3,(H,44,47)
InChI 密鑰
MBDBAPHHKCSIAT-UHFFFAOYSA-N
應用
其他說明
Portal: Building PROTAC® Degraders for Targeted Protein Degradation
Covalent Ligand Screening Uncovers a RNF4 E3 Ligase Recruiter for Targeted Protein Degradation Applications
Targeted Protein Degradation by Small Molecules
Small-Molecule PROTACS: New Approaches to Protein Degradation
Targeted Protein Degradation: from Chemical Biology to Drug Discovery
法律資訊
相關產品
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
文章
Protein Degrader Building Blocks 是一系列交聯劑-E3 配體結合物,具有懸垂功能基團,可與目標配體共價連結。
Protein Degrader Building Blocks are a collection of crosslinker-E3 ligand conjugates with a pendant functional group for covalent linkage to a target ligand.
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務


![氯[三(对三氟甲基苯基)膦]金(I) 99%](/deepweb/assets/sigmaaldrich/product/structures/250/453/f96e05ee-0d9c-46a0-b0f5-818f89e15a2e/640/f96e05ee-0d9c-46a0-b0f5-818f89e15a2e.png)