推薦產品
形狀
crystals
品質等級
饋電比
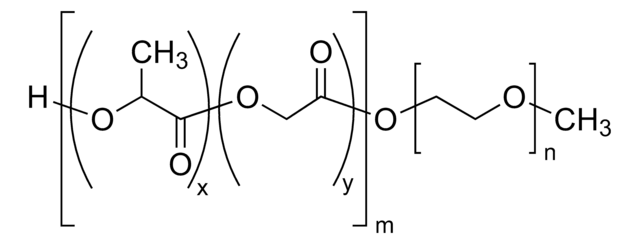
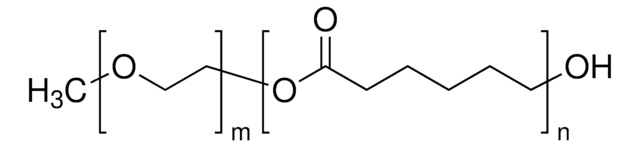
lactide:glycolide 50:50
分子量
PEG average Mn 5,000
PLGA Mn 5,000
雜質
≤5000 ppm (GC)
儲存溫度
−20°C
尋找類似的產品? 前往 產品比較指南
一般說明
通过 GC 分析含有 ≤500 ppm 杂质,包括痕量单体和残留有机物。
應用
生物相容性嵌段共聚物,可用于形成用于药物传递的纳米颗粒具有用于癌症药物、抗炎药、抗生素或麻醉剂的靶向或受控释放的潜力。
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
Fabienne Danhier et al.
Journal of controlled release : official journal of the Controlled Release Society, 133(1), 11-17 (2008-10-28)
The purpose of this study was to develop Cremophor EL-free nanoparticles loaded with Paclitaxel (PTX), intended to be intravenously administered, able to improve the therapeutic index of the drug and devoid of the adverse effects of Cremophor EL. PTX-loaded PEGylated
Miles A Miller et al.
Nature communications, 6, 8692-8692 (2015-10-28)
Therapeutic nanoparticles (TNPs) aim to deliver drugs more safely and effectively to cancers, yet clinical results have been unpredictable owing to limited in vivo understanding. Here we use single-cell imaging of intratumoral TNP pharmacokinetics and pharmacodynamics to better comprehend their
Yihan Xu et al.
Journal of biomedical materials research. Part B, Applied biomaterials, 105(6), 1692-1716 (2016-04-22)
Poly (lactic-co-glycolic acid) (PLGA) copolymers have been broadly used in controlled drug release applications. Because these polymers are biodegradable, they provide an attractive option for drug delivery vehicles. There are a variety of material, processing, and physiological factors that impact
R Gref et al.
Science (New York, N.Y.), 263(5153), 1600-1603 (1994-03-18)
Injectable nanoparticulate carriers have important potential applications such as site-specific drug delivery or medical imaging. Conventional carriers, however, cannot generally be used because they are eliminated by the reticulo-endothelial system within seconds or minutes after intravenous injection. To address these
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務