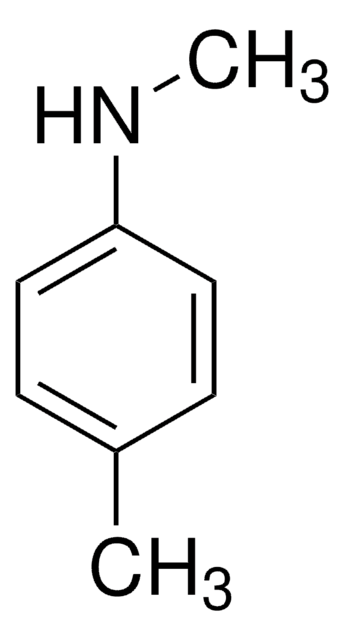
467278
N-甲基-邻甲苯胺
≥95%
同義詞:
2,N-Dimethylaniline, 2-Methyl-N-methylaniline, N,2-Dimethylaniline, N,2-Dimethylbenzenamine, N,o-Dimethylaniline, N-(2-Methylphenyl)methylamine, N-Methyl-2-methylaniline, N-Methyl-2-methylbenzenamine, N-Methyl-N-(o-tolyl)amine, N-Methyl-o-methylaniline, o,N-Dimethylaniline, Methyl(o-tolyl)amine, NSC 9395
About This Item
推薦產品
化驗
≥95%
bp
177-204 °C/740 mmHg (lit.)
密度
0.981 g/mL at 25 °C (lit.)
SMILES 字串
CNc1ccccc1C
InChI
1S/C8H11N/c1-7-5-3-4-6-8(7)9-2/h3-6,9H,1-2H3
InChI 密鑰
GUAWMXYQZKVRCW-UHFFFAOYSA-N
一般說明
應用
- Palladium-catalyzed coupling reactions
- Palladium-catalyzed amination of bromoindazoles
訊號詞
Danger
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Aquatic Chronic 3 - STOT RE 2
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
174.9 °F - closed cup
閃點(°C)
79.4 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務