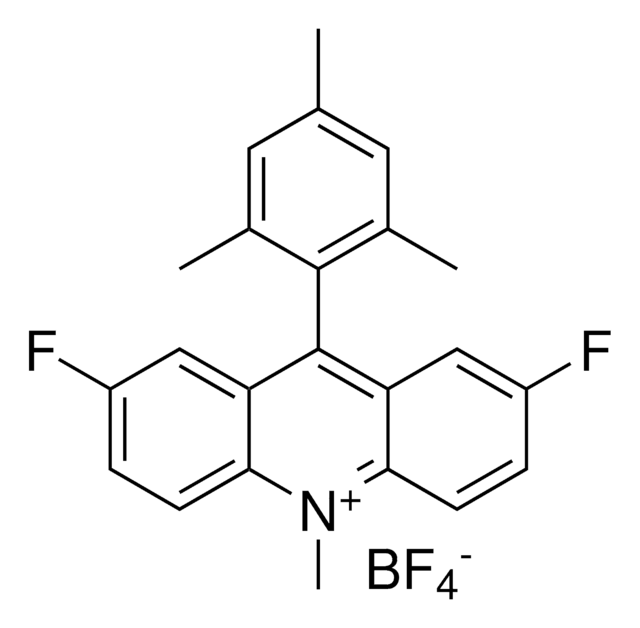
920223
3,6-Di-tert-butyl-9-(2,6-dimethylphenyl)-10-(4-(trifluoromethyl)phenyl)acridin-10-ium tetrafluoroborate
≥95%
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C36H37BF7N
CAS Number:
Molecular Weight:
627.49
UNSPSC Code:
12352103
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥95%
form
powder or crystals
reaction suitability
reaction type: Photocatalysis
reagent type: catalyst
storage temp.
−20°C
Related Categories
Application
3,6-Di-tert-butyl-9-(2,6-dimethylphenyl)-10-(4-(trifluoromethyl)phenyl)acridin-10-ium tetrafluoroborate is a robust acridinium-based photocatalyst reported by Nicewicz and coworkers as an alternative to transition-metal-based photocatalysts with higher chemical stability and attenuated redox potential.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Vincent A Pistritto et al.
Journal of the American Chemical Society, 142(40), 17187-17194 (2020-09-29)
Nucleophilic aromatic substitution (SNAr) is a classical reaction with well-known reactivity toward electron-poor fluoroarenes. However, electron-neutral and electron-rich fluoro(hetero)arenes are considerably underrepresented. Herein, we present a method for the nucleophilic defluorination of unactivated fluoroarenes enabled by cation radical-accelerated nucleophilic aromatic
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service