673862
2-Nitrophenylboronic acid
≥95%
Synonym(s):
o-Nitrobenzeneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
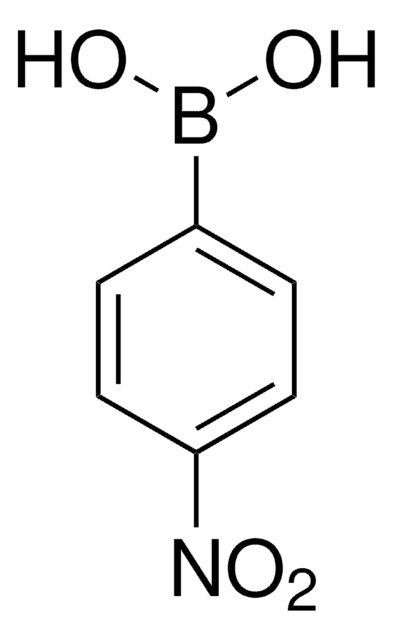
Linear Formula:
(O2N)C6H4(B(OH)2)
CAS Number:
Molecular Weight:
166.93
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥95%
form
powder
mp
155-160 °C
SMILES string
OB(O)c1ccccc1[N+]([O-])=O
InChI
1S/C6H6BNO4/c9-7(10)5-3-1-2-4-6(5)8(11)12/h1-4,9-10H
InChI key
SFUIGUOONHIVLG-UHFFFAOYSA-N
Application
Boronic acid catalyst used for:
Reactant involved in:
- Mild and selective dipolar cycloadditions of unsaturated carboxylic acids with azides
- Transposition of allylic alcohols and Meyer-Schuster rearrangements
Reactant involved in:
- Suzuki-Miyaura cross-coupling reactions with aryl halides or nitroarenediazonium tetrafluoroborates
- Copper-catalyzed halogenation
- Oxidative arylation of aminopyrazolyl disulfides
Used in a preparation of unsymmetrical 2,2′-dinitrobiphenyls via Suzuki-Miyaura cross-coupling with nitrophenyl halides.
Other Notes
May contain varying amounts of anhydride
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Raquel Rodríguez González et al.
The Journal of organic chemistry, 70(23), 9591-9594 (2005-11-05)
[Reaction: see text]. Mechanistic investigations and protocols for the synthesis of 2-nitrobiphenyls and 2,2'-dinitrobiphenyls are disclosed. It is revealed that obstacles appear during the transmetalation step when the phenylboronic acid is substituted with a nitro group in the 2-position, whereas
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)