All Photos(1)
About This Item
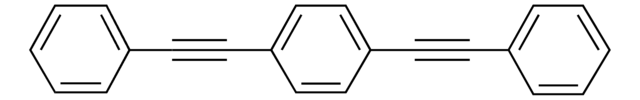
Linear Formula:
C6H5C≡CC≡CC6H5
CAS Number:
Molecular Weight:
202.25
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
mp
86-87 °C (lit.)
SMILES string
c1ccc(cc1)C#CC#Cc2ccccc2
InChI
1S/C16H10/c1-3-9-15(10-4-1)13-7-8-14-16-11-5-2-6-12-16/h1-6,9-12H
InChI key
HMQFJYLWNWIYKQ-UHFFFAOYSA-N
General description
1,4-Diphenylbutadiyne reacts with [WI2(CO)3(NCMe)2] in CH2Cl2 to give the iodo-bridged dimer [W(μ-I)I(CO)(NCMe)(η2PhC2C2Ph)]2. 1,4-Diphenylbutadiyne on UV irradiation with olefins such as 2,3-dimethyl-2-butene, 1,4-cyclohexadiene and dimethyl fumarate yields cross-cycloaddition products.
Application
1,4-Diphenylbutadiyne was used in the preparation of 7,8-dehydropurpurin dimers via two-fold Pd-catalyzed [3+2] annulation of meso-bromoporphyrin.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
1,4-Diphenylbutadiyne, a new non-photodynamic phototoxic compound.
J Kagan et al.
Photochemistry and photobiology, 32(5), 607-611 (1980-11-01)
Carsten Glock et al.
Chemical communications (Cambridge, England), 48(56), 7094-7096 (2012-06-12)
Calciate-mediated intermolecular hydroamination of diphenylbutadiyne with N-phenyl and N-isopropyl-substituted anilines yields E- and Z-isomers of the corresponding 1-anilino-1,4-diphenylbut-1-ene-3-yne. In the case of HNPh(2) solely heterobimetallic K(2)Ca(NPh(2))(4) is able to effectively catalyze this hydroamination reaction in tetrahydrofuran at elevated temperatures.
1, 4-Diphenylbutadiyne complexes of tungsten (II).
Ajayi-Obe T, et al.
Journal of Organometallic Chemistry, 468(1), 165-170 (1994)
Photoaddition reactions of 1, 4-diphenylbutadiyne with olefins.
Sang CS and Sung SK.
Tetrahedron Letters, 26(6), 765-766 (1985)
Norihito Fukui et al.
Angewandte Chemie (International ed. in English), 53(17), 4395-4398 (2014-03-20)
7,8-Dehydropurpurin has attracted much attention owing to the dual 18π- and 20π-electron circuits in its macrocyclic conjugation. The two-fold Pd-catalyzed [3+2] annulation of meso-bromoporphyrin with 1,4-diphenylbutadiyne furnished 7,8-dehydropurpurin dimers. The 8(a) ,8(a) -linked dimer displays a red-shifted and enhanced absorption
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service