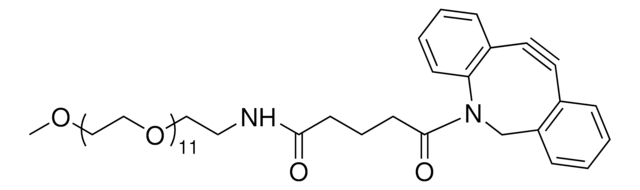
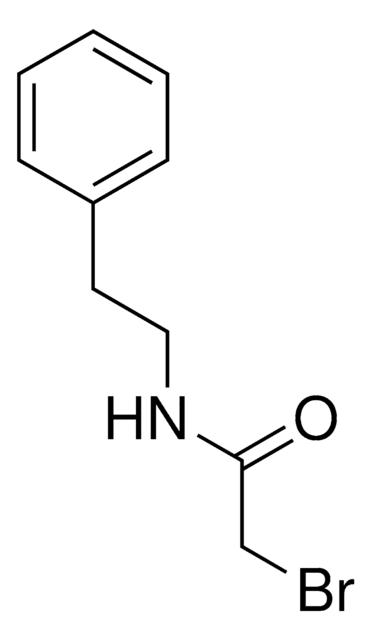
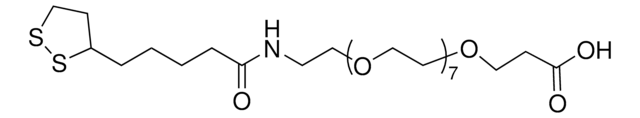
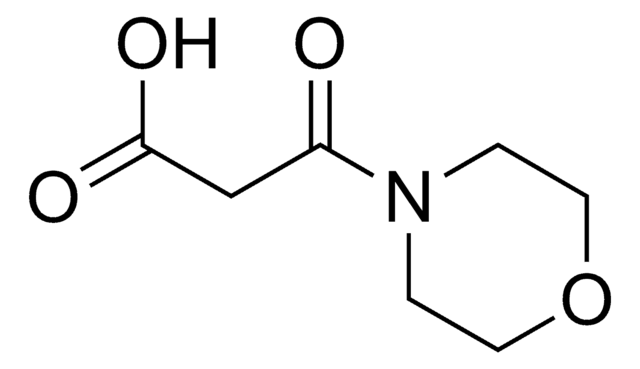
推荐产品
方案
≥95% (LC/MS-ELSD)
表单
solid
应用
metabolomics
vitamins, nutraceuticals, and natural products
储存温度
−20°C
SMILES字符串
N(CCc1ccc(cc1)O)C(=O)C
InChI
1S/C10H13NO2/c1-8(12)11-7-6-9-2-4-10(13)5-3-9/h2-5,13H,6-7H2,1H3,(H,11,12)
InChI key
ATDWJOOPFDQZNK-UHFFFAOYSA-N
一般描述
Natural product derived from fungal source.
警示用语:
Danger
危险声明
危险分类
Eye Dam. 1
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
历史批次信息供参考:
W S Garcez et al.
Journal of agricultural and food chemistry, 48(8), 3662-3665 (2000-08-24)
Two saprophytic fungi (Mucor ramosissimus and Rhizopus sp.) were tested for their ability to induce phytoalexin production by seeds of frog-eye leaf spot and stem canker-resistant and -susceptible soybean (Glycine max L.) cultivars. Only M. ramosissimus was shown to elicit
P H Yu et al.
Canadian journal of biochemistry, 57(10), 1204-1209 (1979-10-01)
The N-acylation of tyramine isomers and other biogenic amines has been studied. The liver exhibits the highest activity towards tyramines, while the brain exhibits a low but significant activity. In the brain, tyramine N-acylation activity was heterogenously distributed. The arylamine
S Friström et al.
Acta pharmacologica et toxicologica, 40(2), 247-258 (1977-02-01)
The effect of five sympathomimetic amines and some of their acetyl derivatives on the blood pressure of the rat was determined on the left carotid artery. After pretreatment with chlorisondamine (1 mg/kg subcutaneously) the blood pressure rise by sympathomimetic amines
A K Mir et al.
Journal of neurochemistry, 36(2), 441-446 (1981-02-01)
N-Acetyltyramine, N-acetyldopamine and N-acetyloctopamine were the major products when either L-[3H]tyrosine or [3H]tyramine were incubated with thoracic ganglia of the desert locust, Schistocerca gregaria. No label was incorporated into L-DOPA under these conditions, although 2-3% of the radioactivity could be
R G Macfarlane et al.
Journal of chromatography, 532(1), 13-25 (1990-10-26)
The N-acetylated metabolites of p-tyramine, p-octopamine and dopamine were identified unambiguously and quantitatively determined in a single ventral thoracic nerve cord of the locust, Schistocerca gregaria, by gas chromatography-negative-ion chemical ionisation mass spectrometry (GC-NICIMS). Deuterium-labelled analogues of each compound were
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门