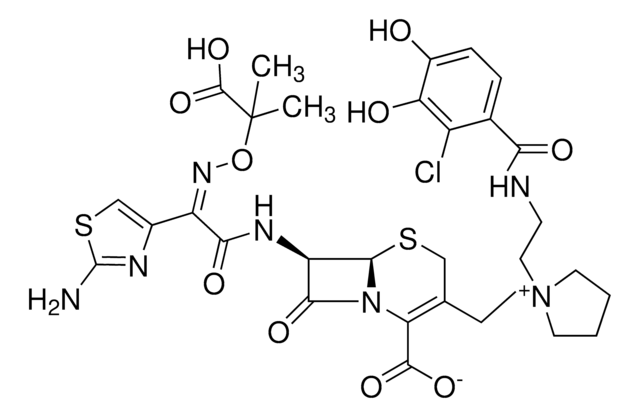
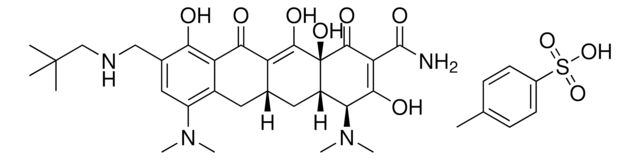
推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
抗生素活性譜
Gram-negative bacteria
Gram-positive bacteria
作用方式
protein synthesis | interferes
儲存溫度
−20°C
InChI
1S/C29H40N4O7/c1-28(2,3)12-31-11-14-10-17(32(4)5)15-8-13-9-16-21(33(6)7)24(36)20(27(30)39)26(38)29(16,40)25(37)18(13)23(35)19(15)22(14)34/h10,13,16,21,31,34,36-37,40H,8-9,11-12H2,1-7H3,(H2,30,39)/t13-,16-,21-,29-/m0/s1
InChI 密鑰
JEECQCWWSTZDCK-IQZGDKDPSA-N
一般說明
Omadacycline is an aminomethylcycline, a novel semisynthetic tetracycline-class antibiotic drug.(1)(2) It is distinguished from other tetracycline-class drugs like tigecycline and eravacycline at the C9 position with an aminomethyl substitution.(2)
應用
Mode of Action: Interferes with protein synthesis Antibiotic Activity Spectrum: Gram-negative bacteria and Gram-positive bacteria.
生化/生理作用
Omadacycline displays antimicrobial activities against a broad spectrum of organisms including Gram-positive, Gram-negative, anaerobic, and atypical bacteria.(3)(4) It inhibits bacterial protein synthesis by binding on the 30S subunit of the bacterial ribosome.(1)(2) Omadacycline is also effective against tetracycline efflux proteins in in vitro studies and protects against bacterial tetracycline resistance.(4) It has improved efficacy than other tetracycline drugs against acute bacterial skin and skin structure infections (ABSSSIs) and community-acquired bacterial pneumonia (CABP).(1)(2)(5) In nonclinical and ex vivo studies, omadacycline displays a low potential for cardiovascular toxicity.(6)
訊號詞
Warning
危險聲明
危險分類
Lact. - Repr. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Rodrigo M Burgos et al.
Infection and drug resistance, 12, 1895-1915 (2019-07-17)
Tetracyclines have come a long way since they became available almost seven decades ago, with numerous enhancements allowing new agents to overcome bacterial mechanisms of resistance. However, these enhancements come with toxicities and pharmacokinetic disadvantages such as the gastrointestinal side-effects
Monique R Bidell et al.
Pharmacotherapy, 41(11), 915-931 (2021-09-25)
Oral tetracyclines have been used in clinical practice for over 60 years. One of the most common indications for use of oral tetracyclines is for treatment of adult outpatients with skin and soft infections (SSTIs), including acute bacterial skin and skin
S Ken Tanaka et al.
Bioorganic & medicinal chemistry, 24(24), 6409-6419 (2016-07-30)
Omadacycline is novel, aminomethyl tetracycline antibiotic being developed for oral and intravenous (IV) administration for the treatment of community-acquired bacterial infections. Omadacycline is characterized by an aminomethyl substituent at the C9 position of the core 6-member ring. Modifications at this
S Ken Tanaka et al.
Antimicrobial agents and chemotherapy, 60(9), 5247-5253 (2016-06-22)
Omadacycline is a first-in-class aminomethylcycline antibiotic with a broad spectrum of activity against Gram-positive and Gram-negative aerobes and anaerobes and atypical bacterial pathogens. A series of nonclinical studies, including mammalian pharmacologic receptor binding studies, human ether-a-go-go-related gene (hERG) channel binding
James A Karlowsky et al.
Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 69(Suppl 1), S6-S15 (2019-08-02)
Omadacycline is a novel aminomethylcycline antimicrobial and semisynthetic derivative of tetracycline. In vitro, omadacycline displays potent activity against gram-positive and many gram-negative bacteria, including methicillin-resistant Staphylococcus aureus, Streptococcus pneumoniae, β-hemolytic streptococci, vancomycin-resistant Enterococcus, and Enterobacteriaceae. Omadacycline is also active against
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门