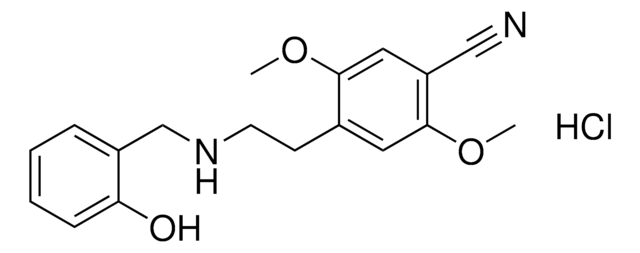
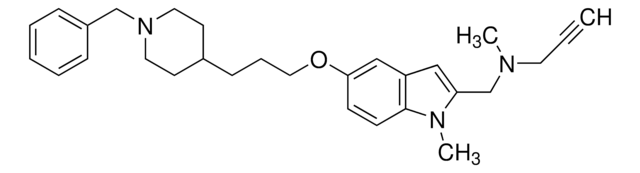
PZ0408
WAY-163909 hydrochloride
≥98% (HPLC)
别名:
(7bR,10aR)-1,2,3,4,7a,9,10,10a-Octahydro-8H-cyclopenta[4,5]pyrrolo[3,2,1-jk][1,4]benzodiazepine, hydrochloride, (7bR,10aR)-1,2,3,4,8,9,10,10a-Octahydro-7bH-cyclopenta-[b][1,4]diazepino[6,7,1hi]indole, hydrochloride, WAY 163909 hydrochloride, WAY-162545 active isomer, WAY-909 hydrochloride, WAY163909 hydrochloride
About This Item
推荐产品
质量水平
方案
≥98% (HPLC)
表单
powder
储存条件
desiccated
颜色
white to beige
溶解性
H2O: 2 mg/mL, clear
储存温度
room temp
SMILES字符串
Cl.N21[C@H]3[C@H](CCC3)c4c2c(ccc4)CNCC1
InChI
1S/C14H18N2.ClH/c1-3-10-9-15-7-8-16-13-6-2-4-11(13)12(5-1)14(10)16;/h1,3,5,11,13,15H,2,4,6-9H2;1H/t11-,13-;/m1./s1
InChI key
UJVFYZZBCUXMDV-LOCPCMAASA-N
生化/生理作用
储存分类代码
11 - Combustible Solids
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门