推荐产品
等級
pharmaceutical primary standard
API 家族
tenoxicam
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
SMILES 字串
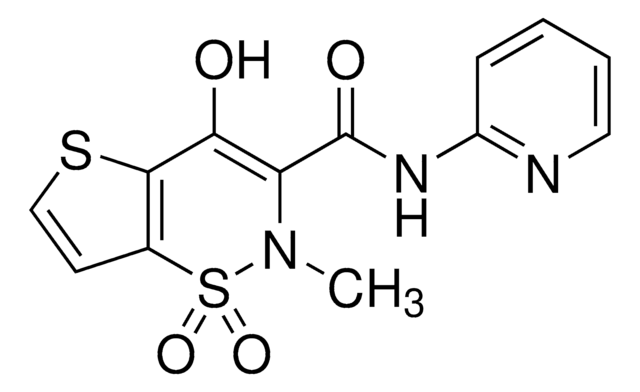
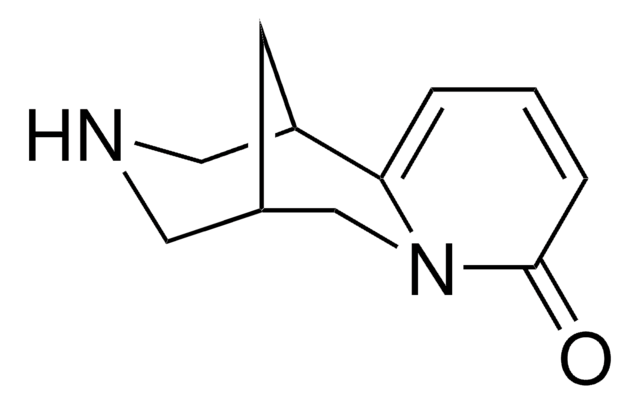
O=C(NC1=NC=CC=C1)C2=C(O)C(SC=C3)=C3S(N2C)(=O)=O
InChI
1S/C13H11N3O4S2/c1-16-10(13(18)15-9-4-2-3-6-14-9)11(17)12-8(5-7-21-12)22(16,19)20/h2-7,17H,1H3,(H,14,15,18)
InChI 密鑰
LZNWYQJJBLGYLT-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Tenoxicam EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
生化/生理作用
Non-steroidal antiinflammatory drug (NSAID) with comparatively low risk of renal or hepatic toxicity.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
訊號詞
Danger
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Demiana I Nesseem et al.
Life sciences, 89(13-14), 430-438 (2011-07-23)
The purpose of this study was to develop transdermal films (TFs) with the addition of different polymer ratios that incorporated 0.5% tenoxicam in order to ensure maximum controlled and sustained drug release capacity. Tenoxicam is a non-steroidal anti-inflammatory drug (NSAID)
I Aktas et al.
International journal of oral and maxillofacial surgery, 39(5), 440-445 (2010-03-10)
This study examined the clinical and radiological effects of intra-articular tenoxicam injection following arthrocentesis and compared them with arthrocentesis alone in patients with disc displacement without reduction (DDwoR). 24 temporomandibular joints (TMJs) in 21 patients with DDwoR were studied. Patients
Owen A Moore et al.
The Cochrane database of systematic reviews, (3)(3), CD007591-CD007591 (2009-07-10)
Tenoxicam is a non-steroidal anti-inflammatory drug (NSAID) licensed for use in rheumatic disease and other musculoskeletal disorders in the UK, and is widely available in other countries worldwide. This review sought to evaluate the efficacy and safety of oral tenoxicam
Berker Cemil et al.
European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society, 20(8), 1255-1258 (2011-02-18)
Post laminectomy arachnoiditis has been shown by experiments with rats and post operative radiological imaging in humans. The purpose of this experimental study was to determine the efficacy of tenoxicam in preventing arachnoiditis in rats. Twenty-four Wistar rats were divided
I Gunusen et al.
Clinical and experimental obstetrics & gynecology, 39(1), 49-52 (2012-06-09)
The purpose of this study was to determine the analgesic efficacy and side-effects of paracetamol and tenoxicam in comparison with placebo in patients with postoperative pain after elective abdominal hysterectomy. A total of 120 patients were randomly divided into three
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门