推荐产品
等級
certified reference material
pharmaceutical secondary standard
品質等級
agency
traceable to Ph. Eur. Y0000633
traceable to USP 1097636
API 家族
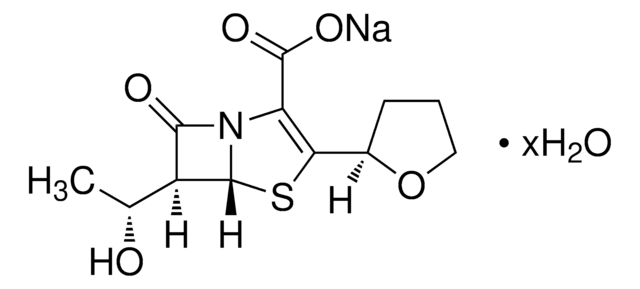
cefepime
CofA
current certificate can be downloaded
包裝
pkg of 1 g
技術
HPLC: suitable
gas chromatography (GC): suitable
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
InChI
1S/C19H24N6O5S2.2ClH.H2O/c1-25(5-3-4-6-25)7-10-8-31-17-13(16(27)24(17)14(10)18(28)29)22-15(26)12(23-30-2)11-9-32-19(20)21-11;;;/h9,13,17H,3-8H2,1-2H3,(H3-,20,21,22,26,28,29);2*1H;1H2/b23-12-;;;/t13-,17-;;;/m1.../s1
InChI 密鑰
LRAJHPGSGBRUJN-OMIVUECESA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Cefepime hydrochloride is one of the fourth-generation, semisynthetic, broad-spectrum, cephalosporin β-lactam antibiotic drugs administered parenterally in patients with moderate-to-severe infections like pneumonia, uncomplicated urinary tract infections, skin and soft tissue infections, etc. Its mode of action involves the penetration of outer cell wall of gram negative bacteria.
Pharmaceutical secondary standard for application in quality control. Provides pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Pharmaceutical secondary standard for application in quality control. Provides pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
應用
Cefepime hydrochloride has been used as a pharmaceutical reference standard for the determination of the analyte in pharmaceutical formulations by spectrophotometry and chromatography techniques.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
分析報告
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
腳註
To see an example of a Certificate of Analysis for this material enter LRAB8503 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
相關產品
产品编号
说明
价格
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Spectrophotometric determination of cefepime hydrochloride, cefoperazone sodium, ceftazidime pentahydrate. cefuroxime sodium and etamsylate using ammonium molybdate
Elazazy M, et al.
Scientia Pharmaceutica, 71(3), 211-228 (2003)
Analysis of N-methylpyrrolidine in cefepime hydrochloride by ion chromatography using suppressed conductivity detection with solid-phase extraction pre-treatment
Page N, et al.
Analytical Methods for Therapeutic Drug Monitoring and Toxicology, 6(4), 1248-1253 (2014)
Development and validation of a green analytical method of RP-HPLC for quantification of Cefepime hydrochloride in pharmaceutical dosage form: Simple, sensitive and economic
Rodrigues DF and Salgado HRN
Current Pharmaceutical Analysis, 12(4), 306-314 (2016)
RP-HPLC method for simultaneous estimation of cefepime hydrochloride and tazobactam sodium in bulk and pharmaceuticals
Tamboli SR and Patil DD
Journal of Chemistry, 2013(4), 1248-1253 (2013)
Lokender Kumar et al.
Frontiers in microbiology, 12, 598498-598498 (2021-02-16)
Pseudomonas aeruginosa utilizes the quorum sensing (QS) system to strategically coordinate virulence and biofilm formation. Targeting QS pathways may be a potential anti-infective approach to treat P. aeruginosa infections. In the present study, we define cephalosporins' anti-QS activity using Chromobacterium
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门