推荐产品
等級
pharmaceutical primary standard
API 家族
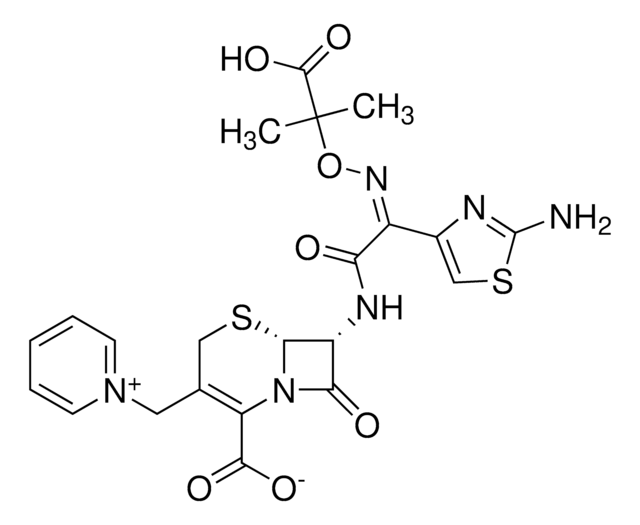
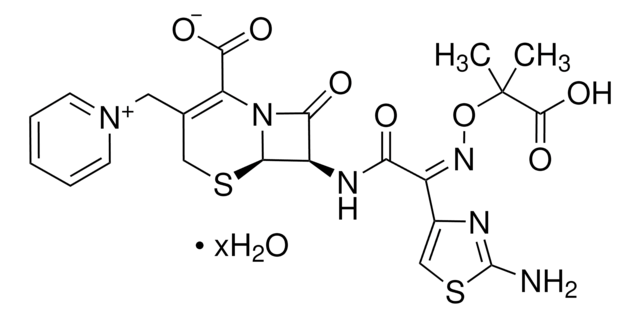
ceftazidime
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
格式
neat
儲存溫度
2-8°C
InChI
1S/C22H22N6O7S2.5H2O/c1-22(2,20(33)34)35-26-13(12-10-37-21(23)24-12)16(29)25-14-17(30)28-15(19(31)32)11(9-36-18(14)28)8-27-6-4-3-5-7-27;;;;;/h3-7,10,14,18H,8-9H2,1-2H3,(H4-,23,24,25,29,31,32,33,34);5*1H2/b26-13-;;;;;/t14-,18-;;;;;/m1...../s1
InChI 密鑰
NMVPEQXCMGEDNH-TZVUEUGBSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Ceftazidime EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
訊號詞
Danger
危險聲明
危險分類
Resp. Sens. 1 - Skin Sens. 1
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Anu Kantele et al.
Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 60(6), 837-846 (2015-01-24)
More than 300 million travelers visit regions with poor hygiene annually. A significant percentage of them become colonized by resistant intestinal bacteria such as extended-spectrum beta-lactamase-producing Enterobacteriaceae (ESBL-PE) and may transmit the strains to others and to medical care settings
Johanna Berkhout et al.
Antimicrobial agents and chemotherapy, 59(2), 1138-1144 (2014-12-10)
To evaluate the in vitro effects of the combination of ceftazidime and avibactam on the MICs of both compounds, checkerboard assays were performed for 81 clinical strains, including 55 Enterobacteriaceae strains (32 Klebsiella pneumoniae, 19 Escherichia coli, 1 Citrobacter freundii
George G Zhanel et al.
Drugs, 73(2), 159-177 (2013-02-02)
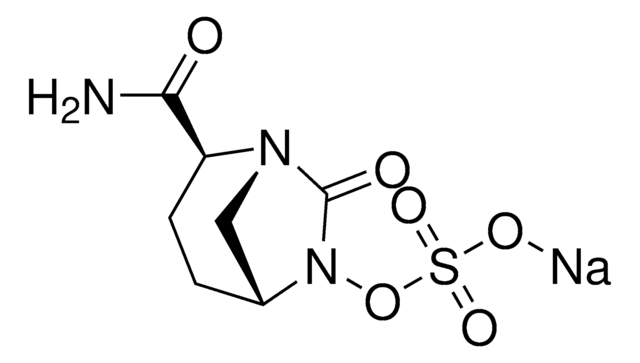
Avibactam (formerly NXL104, AVE1330A) is a synthetic non-β-lactam, β-lactamase inhibitor that inhibits the activities of Ambler class A and C β-lactamases and some Ambler class D enzymes. This review summarizes the existing data published for ceftazidime-avibactam, including relevant chemistry, mechanisms
A Walkty et al.
Antimicrobial agents and chemotherapy, 57(11), 5707-5709 (2013-08-14)
The in vitro activity of ceftolozane in combination with tazobactam (fixed concentration of 4 μg/ml) was evaluated against 2,435 Pseudomonas aeruginosa clinical isolates obtained from across Canada using Clinical and Laboratory Standards Institute broth microdilution methods. The MIC50 and MIC90
Gavin C K W Koh et al.
PLoS neglected tropical diseases, 7(10), e2500-e2500 (2013-10-23)
Burkholderia pseudomallei infection (melioidosis) is an important cause of community-acquired Gram-negative sepsis in Northeast Thailand, where it is associated with a ~40% mortality rate despite antimicrobial chemotherapy. We showed in a previous cohort study that patients taking glyburide ( =
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门