推荐产品
品質等級
化驗
≥95%
形狀
powder or crystals
反應適用性
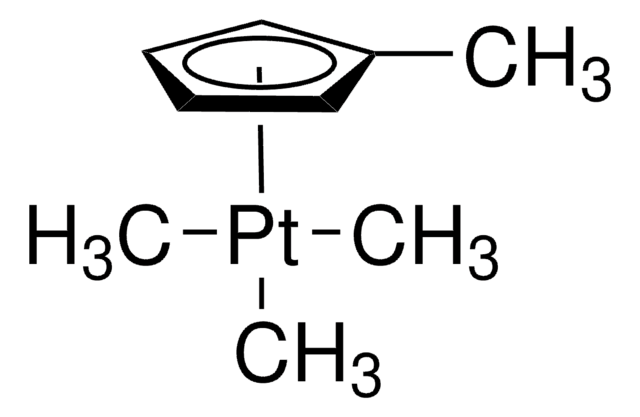
reaction type: C-C Bond Formation
reagent type: catalyst
reagent type: ligand
reaction type: C-H Activation
應用
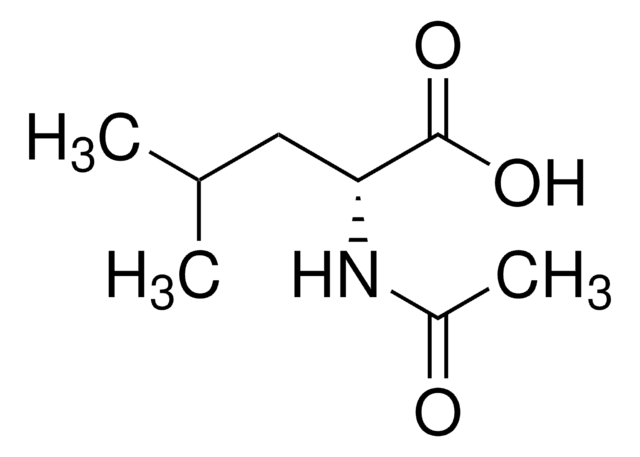
This chiral acetyl-protected aminoethyl quinoline (APAQ) ligand was developed by the Yu group for ligand-enabled acceleration of Pd-catalyzed enantioselective β-methylene C–H functionalization, which is important for the generation of β-chiral centers in asymmetric synthesis.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
相关内容
The Yu program centers around the discovery of catalytic carbon–carbon and carbon–heteroatom bond forming reactions based on C–H activation. Target transformations are selected to enable 1) the use of simple and abundant starting materials such as aliphatic acids, amines and alcohols, and 2) disconnections that drastically shorten the synthesis of a drug molecule or a major class of biologically active compounds.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门