所有图片(3)
选择尺寸
变更视图
1 G
$234.00
5 G
$1,040.00
About This Item
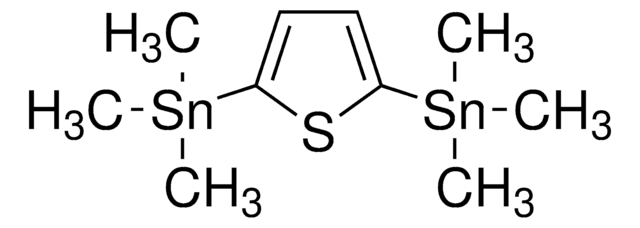
经验公式(希尔记法):
C12H20S2Sn2
分子量:
465.84
MDL编号:
UNSPSC代码:
12352103
PubChem化学物质编号:
NACRES:
NA.23
推荐产品
质量水平
方案
97%
表单
powder or crystals
mp
127-132 °C
SMILES字符串
C[Sn](C)(C)c1cc2sc(cc2s1)[Sn](C)(C)C
InChI
1S/C6H2S2.6CH3.2Sn/c1-3-7-6-2-4-8-5(1)6;;;;;;;;/h1-2H;6*1H3;;
InChI key
HDZULVYGCRXVNQ-UHFFFAOYSA-N
一般描述
应用
2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene can be used as a copolymer in the synthesis of thiophene based materials for the fabrication of organic electronic devices such as organic field effect transistors (OFETs), organic thin film transistors(OTFTs) and organic photovoltaic cells (OPVs).[3][4]
2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene is a synthetic intermediate, which can be used in the synthesis of polythiophenes based fluorinated polymers for the optoelectronic applications.[5] It can also be used in the synthesis of PDBT-co-TT via Still coupling polymerization for the fabrication of organic thin film transistors (OTFTs)and photovoltaic devices.[2][3][1]
警示用语:
Danger
危险分类
Acute Tox. 1 Dermal - Acute Tox. 2 Inhalation - Acute Tox. 2 Oral - Aquatic Acute 1 - Aquatic Chronic 1
储存分类代码
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
Dialkyl-Substituted Thieno [3, 2-b] thiophene-Based Polymers Containing 2, 2 `-Bithiophene, Thieno [3, 2-b] thiophene, and Ethynylene Spacers
San Miguel L and Matzger AJ
Macromolecules, 40(26), 9233-9237 (2007)
Thienoisoindigo-based copolymer with fused thieno (3, 2-b) thiophene as a donor in thin film transistor applications with high performance
Chen C, et al.
Journal of Material Chemistry C, 3(1), 33-36 (2015)
A High Mobility P-Type DPP-Thieno [3, 2-b] thiophene Copolymer for Organic Thin-Film Transistors
Li Y, et al.
Advanced Materials, 22(43), 4862-4866 (2010)
Using Molecular Design to Increase Hole Transport: Backbone Fluorination in the Benchmark Material Poly (2, 5-bis (3-alkylthiophen-2-yl) thieno [3, 2-b]-thiophene (pBTTT)
Boufflet P, et al.
Advances in Functional Materials, 25(45), 7038-7048 (2015)
Hugo Bronstein et al.
Journal of the American Chemical Society, 133(10), 3272-3275 (2011-02-22)
We report the synthesis and polymerization of a novel thieno[3,2-b]thiophene-diketopyrrolopyrrole-based monomer. Copolymerization with thiophene afforded a polymer with a maximum hole mobility of 1.95 cm(2) V(-1) s(-1), which is the highest mobility from a polymer-based OFET reported to date. Bulk-heterojunction
商品
Organic electronics promise renewable energy solutions surpassing silicon-based tech.
Active Filters
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持![1,1′-[4,8-Bis[5-(2-ethylhexyl)-2-thienyl]benzo[1,2-b:4,5-b′]dithiophene-2,6-diyl]bis[1,1,1-trimethylstannane]](/deepweb/assets/sigmaaldrich/product/structures/611/912/a638a6fe-ca7b-4674-8023-df4c0921a9fd/640/a638a6fe-ca7b-4674-8023-df4c0921a9fd.png)



![苯并[1,2B:4,5-B]二噻吩-4- -1,8-二酮 97%](/deepweb/assets/sigmaaldrich/product/structures/418/544/b7faac0b-ad09-4b42-a9fa-aeb38017a39e/640/b7faac0b-ad09-4b42-a9fa-aeb38017a39e.png)
![噻吩并[3,2-b]噻吩 95%](/deepweb/assets/sigmaaldrich/product/structures/353/609/429fd4bf-e217-4371-80a3-9e5a4d88908b/640/429fd4bf-e217-4371-80a3-9e5a4d88908b.png)


![[1,1′-双(二苯基膦)二茂铁]二氯化钯(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)

