B7273
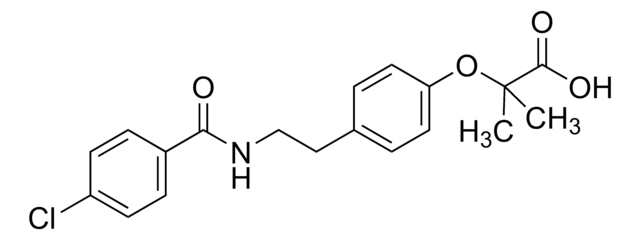
Bezafibrate
≥98%, solid
Synonym(s):
2-[4-[2-(4-Chlorobenzamido)ethyl]phenoxy]-2-methylpropanoic acid
About This Item
Recommended Products
Assay
≥98%
form
solid
solubility
DMF: soluble
deionized water: insoluble
methanol: soluble
originator
Roche
SMILES string
CC(C)(Oc1ccc(CCNC(=O)c2ccc(Cl)cc2)cc1)C(O)=O
InChI
1S/C19H20ClNO4/c1-19(2,18(23)24)25-16-9-3-13(4-10-16)11-12-21-17(22)14-5-7-15(20)8-6-14/h3-10H,11-12H2,1-2H3,(H,21,22)(H,23,24)
InChI key
IIBYAHWJQTYFKB-UHFFFAOYSA-N
Gene Information
human ... HBA2(3040) , PPARA(5465) , PPARD(5467) , PPARG(5468)
mouse ... Ppara(19013) , Ppard(19015) , Pparg(19016)
Looking for similar products? Visit Product Comparison Guide
Application
- a supplement in the standard diet (SD) for mice to study its effect on diabetes
- to evaluate its effect on hepatitis C virus (HCV) assembly and secretion
- to evaluate its effect on gonadal steroidogenesis and spermatogenesis of zebrafish and also used as standard for HPLC
Biochem/physiol Actions
PPARgamma agonists, including Bezafibrate, have beneficial effects in the suppression of the inflammatory response during RSV infection and therefore might have clinical efficacy in the course of severe RSV-infection.
Features and Benefits
Signal Word
Warning
Hazard Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We offer a variety of small molecule research tools, such as transcription factor modulators, inhibitors of chromatin modifying enzymes, and agonists/antagonists for target identification and validation in gene regulation research; a selection of these research tools is shown below.
We offer a variety of small molecule research tools, such as transcription factor modulators, inhibitors of chromatin modifying enzymes, and agonists/antagonists for target identification and validation in gene regulation research; a selection of these research tools is shown below.
We offer a variety of small molecule research tools, such as transcription factor modulators, inhibitors of chromatin modifying enzymes, and agonists/antagonists for target identification and validation in gene regulation research; a selection of these research tools is shown below.
We offer a variety of small molecule research tools, such as transcription factor modulators, inhibitors of chromatin modifying enzymes, and agonists/antagonists for target identification and validation in gene regulation research; a selection of these research tools is shown below.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service