After administration by the mouth, the half-life is 2 to 5 hours 1. In plasma, about 92-98% binds to plasma proteins. Nifedipine is completely metabolized. About 70% of a dose is excreted in the urine in 24 hours as metabolites including 5-methoxycarbonyl-2,6-dimethyl-4-(2-nitrophenyl) pyridine-3-carboxylic acid; dimethyl 2,6-dimethyl-4-(2-nitrophenyl pyridine-3,5-dicarboxylate and 2-hydroxymethyl-5-methoxycarbonyl-6-methyl-4-(2-nitrophenyl) pyridine-3-carboxylic acid and its lactone derivative. Up to 15% of a dose is eliminated in the feces as metabolites in 4 days 2. References: 1. Martindale, 29th ed., pgs. 1509-1513. 2. Clarke's Isolation and Identification of Drugs., 2nd ed., p. 811.
N7634
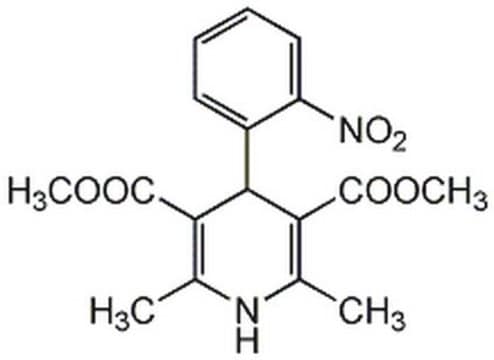
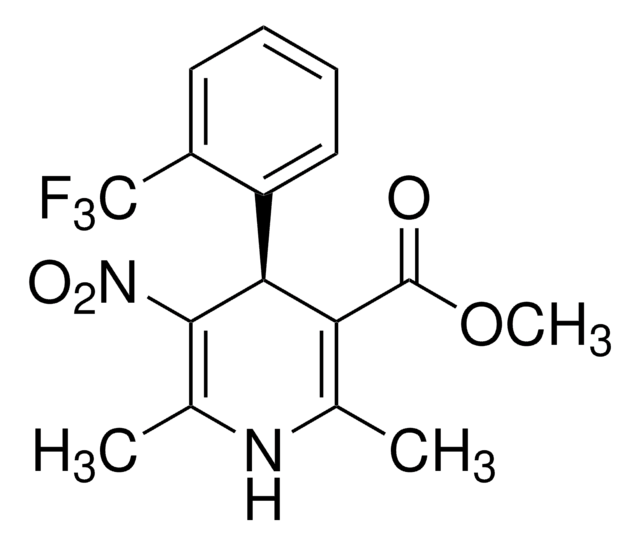
Nifedipine
≥98% (HPLC), powder, L-type Ca²⁺ channel blocker
Sinónimos:
1,4-Dihydro-2,6-dimethyl-4-(2-nitrophenyl)-3,5-pyridinedicarboxylic acid dimethyl ester
About This Item
Productos recomendados
Nombre del producto
Nifedipine, ≥98% (HPLC), powder
Nivel de calidad
Ensayo
≥98% (HPLC)
Formulario
powder
color
yellow
solubilidad
DMSO: soluble
ethanol: soluble
emisor
Bayer
temp. de almacenamiento
2-8°C
cadena SMILES
COC(=O)C1=C(C)NC(C)=C(C1c2ccccc2[N+]([O-])=O)C(=O)OC
InChI
1S/C17H18N2O6/c1-9-13(16(20)24-3)15(14(10(2)18-9)17(21)25-4)11-7-5-6-8-12(11)19(22)23/h5-8,15,18H,1-4H3
Clave InChI
HYIMSNHJOBLJNT-UHFFFAOYSA-N
Información sobre el gen
human ... ADORA2A(135) , ADORA3(140) , CACNA1C(775) , CACNA1D(776) , CACNA1F(778) , CACNA1S(779) , CACNA2D1(781) , CYP1A2(1544) , KCNH1(3756) , TTR(7276)
mouse ... Cacna1c(12288)
rat ... Adora1(29290) , Adora2a(25369) , Cacna1c(24239) , Cacna1d(29716) , Kcnj1(24521) , Kcnn4(65206) , Tbxas1(24886)
¿Está buscando productos similares? Visita Guía de comparación de productos
Aplicación
- to evaluate its effect on myenteric neuronal calcium current through R-type calcium channel in guinea pig small intestine[1]
- to evaluate the neuroprotective effect of L-type calcium channel blockers in cholinergic and dopaminergic neurons[2]
- to identify the effect of co-administration of nifedipine (anti-hypertensive drug) along with hypoglycemic drug on human umbilical vein cells (HUVECs)[3]
Acciones bioquímicas o fisiológicas
Características y beneficios
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 1
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Cyclic nucleotides like cAMP modulate cell function via PKA activation and ion channels.
Cyclic nucleotides like cAMP modulate cell function via PKA activation and ion channels.
Cyclic nucleotides like cAMP modulate cell function via PKA activation and ion channels.
Cyclic nucleotides like cAMP modulate cell function via PKA activation and ion channels.
-
What is the half life of Product N7634, Nifidipine, in vivo?
1 answer-
Helpful?
-
-
What is the Department of Transportation shipping information for this product?
1 answer-
Transportation information can be found in Section 14 of the product's (M)SDS.To access the shipping information for this material, use the link on the product detail page for the product.
Helpful?
-
-
What is the solubility of Product N7634, Nifidipine?
1 answer-
Nifedipine can be dissolved in DMSO at 50 mg/ml1. It is sparingly soluble in absolute ethanol2. Herembert, T. et al., dissolved nifedipine in absolute ethanol (no concentration reported); the maximum ethanol concentration in cultures was 0.2% without any effect of solvent on the cells3. Nifedipine is soluble (g/L, at 20°C) in the following solvents: acetone, 250; methylene chloride, 160; chloroform, 140; ethyl acetate, 50; methanol, 26; ethanol, 17.4 It is practically insoluble in water. The solubilities at 37°C in buffer solutions of different pH values are: pH 4, 0.0058 g/L; pH 7, 0.0056 g/L; pH 9.0, 0.0078 g/L; pH 13, 0.006 g/L1. References: 1. Ali, S.L., Analytical Profiles of Drug Substances, 18, 221, (1989). 2. Martindale, The Extra Pharmacopoeia, 30th ed., 374, (1993). and 3. Herembert, T. et al. Brit. J. Pharmacol., 114, 1703, (1995).
Helpful?
-
-
Are there any recommended dosages for Product N7634, Nifidipine, in vivo and in vitro?
1 answer-
Nifedipine is reported to inhibit Ca2+-sensitive K+ channels at 100 μM1. Doses for different animals have been reported2,3. In randomly growing cultures of aortic cells of rats, nifedipine at 10 μM inhibited cell proliferation. References: 1. Thomas-Young, R.J. et al. Biochim. Biophys. Acta, 1146, 81, (1993). 2. Drug Dosages In Laboratory Animals: A Handbook, 3rd ed., Telford Press. and 3. Borchard, R.E. et al. Drug Dosage in Laboratory Animals, A Handbook, Third Edition, p. 315, The Telford Press, (1990).
Helpful?
-
-
Are there any recommended conditions that Product N7634, Nifidipine, should be used in?
1 answer-
When exposed to daylight and certain wavelengths of artificial light, nifedipine readily converts to a nitrosophenylpyridine derivative. Exposure to ultra-violet light leads to the formation of a nitrophenylpyridine derivative. Therefore, USP recommends that assays be performed in the dark or under golden fluorescent or other low actinic light. Low actinic glassware should be used1.
Helpful?
-
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico