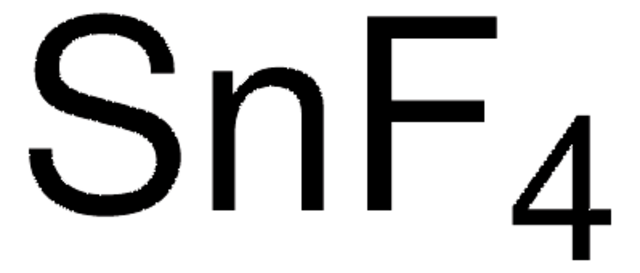14520
Tin(IV) chloride
fuming, ≥99%
Synonym(s):
Stannic chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
SnCl4
CAS Number:
Molecular Weight:
260.52
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
Quality Level
Assay
≥99%
reaction suitability
reagent type: catalyst
core: tin
density
2.217 g/mL at 20 °C (lit.)
anion traces
sulfate (SO42-): ≤100 mg/kg
cation traces
As: ≤50 mg/kg
Fe: ≤10 mg/kg
SMILES string
Cl[Sn](Cl)(Cl)Cl
InChI
1S/4ClH.Sn/h4*1H;/q;;;;+4/p-4
InChI key
HPGGPRDJHPYFRM-UHFFFAOYSA-J
Looking for similar products? Visit Product Comparison Guide
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Evangelina Repetto et al.
Bioorganic & medicinal chemistry, 17(7), 2703-2711 (2009-03-14)
Thiodisaccharides having beta-D-Galf or alpha-L-Araf units as non-reducing end have been synthesized by the SnCl(4)- or MoO(2)Cl(2)-promoted thioglycosylation of per-O-benzoyl-D-galactofuranose (1), its 1-O-acetyl analogue 4, or per-O-acetyl-alpha-L-arabinofuranose (16) with 6-thioglucose or 6-thiogalactose derivatives. After convenient removal of the protecting groups
Yu-Jun Zhao et al.
Chemical communications (Cambridge, England), (12)(12), 1434-1436 (2008-03-14)
This communication describes a highly efficient intermolecular polyene cyclization method using steroidal acetals as the initiators to synthesize tetracyclic terpenoids; both good yields and good asymmetric induction were obtained.
Yu-Jun Zhao et al.
Journal of the American Chemical Society, 130(30), 10024-10029 (2008-07-09)
New strategies using chiral acetal or chiral mixed-acetal in the presence of Lewis acids (SnCl4 or TiCl4) to promote polyene cyclization reaction are described. Acetal-promoted and mixed-acetal-promoted polyene cyclization products are very versatile and can easily be converted into various
Andreas T Messmer et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 18(47), 14989-14995 (2012-10-04)
Determining the structure of reactive intermediates is the key to understanding reaction mechanisms. To access these structures, a method combining structural sensitivity and high time resolution is required. Here ultrafast polarization-dependent two-dimensional infrared (P2D-IR) spectroscopy is shown to be an
Leo A Paquette et al.
The Journal of organic chemistry, 69(22), 7442-7447 (2004-10-23)
Tin tetrachloride-catalyzed glycosidation of persilylated nucleobases with acetate donor 6 in CH(2)Cl(2) solution followed by deprotection gave rise very predominantly to alpha-spironucleosides. These stereochemical assignments stem from the determination of NOE interactions and an X-ray crystallographic analysis of the latter
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service