06627
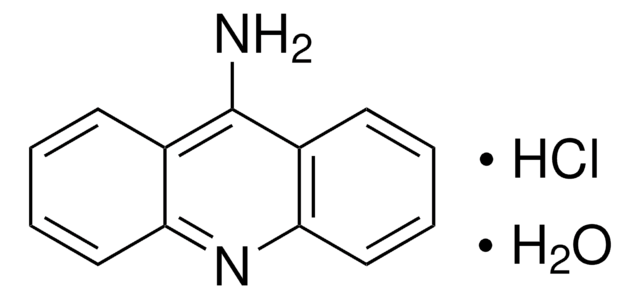
2-Aminoacridone
BioReagent, suitable for fluorescence, ≥98.0% (HPLC)
Sinónimos:
2-Amino-9(10H)-acridinone, 2-amino-10H-acridin-9-one, AMAC
Seleccione un Tamaño
Seleccione un Tamaño
About This Item
Productos recomendados
Línea del producto
BioReagent
Ensayo
≥98.0% (HPLC)
solubilidad
DMF: soluble
DMSO: soluble
fluorescencia
λex 420 nm; λem 542 nm in 0.1 M Tris pH 8.0
idoneidad
suitable for fluorescence
cadena SMILES
Nc1ccc2Nc3ccccc3C(=O)c2c1
Nc1ccc2Nc3ccccc3C(=O)c2c1
InChI
1S/C13H10N2O/c14-8-5-6-12-10(7-8)13(16)9-3-1-2-4-11(9)15-12/h1-7H,14H2,(H,15,16)
Clave InChI
PIGCSKVALLVWKU-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Aplicación
Envase
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Glycans play a key role in protein structure and disease; representation on cell surfaces is the glycome.
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico