All Photos(1)
About This Item
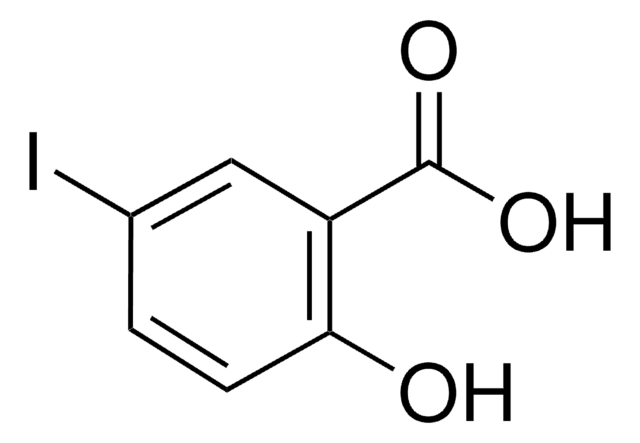
Linear Formula:
2-(HO)C6H4CONH2
CAS Number:
Molecular Weight:
137.14
Beilstein:
742439
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
solid
mp
140-144 °C (lit.)
SMILES string
NC(=O)c1ccccc1O
InChI
1S/C7H7NO2/c8-7(10)5-3-1-2-4-6(5)9/h1-4,9H,(H2,8,10)
InChI key
SKZKKFZAGNVIMN-UHFFFAOYSA-N
Gene Information
human ... AR(367)
Looking for similar products? Visit Product Comparison Guide
General description
Salicylamide is used as a starting material for synthesizing substituted salicylamides through acylation reactions.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Acylation of salicylamide to 5-acetylsalicylamide using ionic liquids as dual catalyst and solvent
Chen, W et al.
Journal of Industrial and Engineering Chemistry, 16(5), 800-804 (2010)
Po-Chiao Lin et al.
Analytical chemistry, 79(9), 3401-3408 (2007-04-04)
Functionalized magnetic nanoparticles (MNPs) were synthesized to serve as laser desorption/ionization elements as well as solid-phase extraction probes for simultaneous enrichment and detection of small molecules in matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) analysis. Two laser-absorbing matrices were
Xiaoyan Zhou et al.
Dalton transactions (Cambridge, England : 2003), 41(6), 1765-1775 (2011-12-14)
Two new flexible exo-bidentate ligands were designed and synthesized, incorporating different backbone chain lengths bearing two salicylamide arms, namely 2,2'-(2,2'-oxybis(ethane-2,1-diyl)bis(oxy))bis(N-benzylbenzamide) (L(I)) and 2,2'-(2,2'-(ethane-1,2-diylbis(oxy))bis(ethane-2,1-diyl))bis(oxy)bis(N-benzylbenzamide) (L(II)). These two structurally related ligands are used as building blocks for constructing diverse lanthanide polymers with
Jessica Nilsson et al.
Journal of inorganic biochemistry, 105(12), 1795-1800 (2011-11-08)
Reaction of N-(2-hydroxybenzyl)-N-(2-picolyl) glycine (H(2)papy) with VOSO(4) in water gives the oxidovanadium(V) oxido-bridged dimer [{(papy)(VO)}(2) μ-O)] (1). Similarly, reaction of N-(2-hydroxybenzyl) glycine (H(2)glysal) with VOSO(4) gives [(glysal)VO(H(2)O)] (2) and reaction of salicylamide (Hsalam) with VOSO(4) in methanol gives [(salam)(2)VO] (3).
Patricia D Sauzem et al.
European journal of medicinal chemistry, 43(6), 1237-1247 (2007-09-25)
In this work, we reported the synthesis and evaluation of the analgesic and anti-inflammatory properties of novel 3- or 4-substituted 5-trifluoromethyl-5-hydroxy-4,5-dihydro-1H-1-carboxyamidepyrazoles (where 3-/4-substituent=H/H, Me/H, Et/H, Pr/H, i-Pr/H, Bu/H, t-Bu/H, Ph/H, 4-Br-Ph/H and H/Me) designed in the exploration of the bioisosteric
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service