推荐产品
产品名称
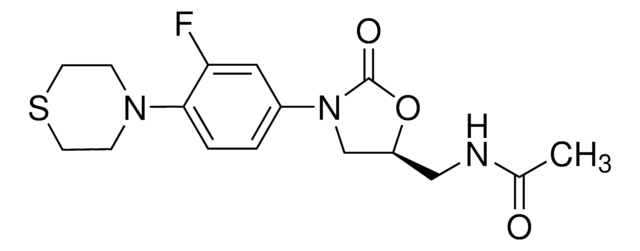
利奈唑胺, ≥98% (HPLC)
方案
≥98% (HPLC)
表单
powder
颜色
white to off-white
溶解性
DMSO: >20 mg/mL
储存温度
room temp
SMILES字符串
CC(=O)NC[C@H]1CN(C(=O)O1)c2ccc(N3CCOCC3)c(F)c2
InChI
1S/C16H20FN3O4/c1-11(21)18-9-13-10-20(16(22)24-13)12-2-3-15(14(17)8-12)19-4-6-23-7-5-19/h2-3,8,13H,4-7,9-10H2,1H3,(H,18,21)/t13-/m0/s1
InChI key
TYZROVQLWOKYKF-ZDUSSCGKSA-N
正在寻找类似产品? 访问 产品对比指南
一般描述
应用
生化/生理作用
警示用语:
Danger
危险声明
危险分类
STOT RE 1 Oral
靶器官
Bone
储存分类代码
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
闪点(°F)
Not applicable
闪点(°C)
Not applicable
历史批次信息供参考:
分析证书(COA)
其他客户在看
商品
Bioactive small molecules for immune system signaling target identification/validation and antibiotics, antivirals, and antifungals offered.
Bioactive small molecules for immune system signaling target identification/validation and antibiotics, antivirals, and antifungals offered.
Bioactive small molecules for immune system signaling target identification/validation and antibiotics, antivirals, and antifungals offered.
Bioactive small molecules for immune system signaling target identification/validation and antibiotics, antivirals, and antifungals offered.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门