推荐产品
化驗
97%
形狀
powder
mp
143-146 °C (lit.)
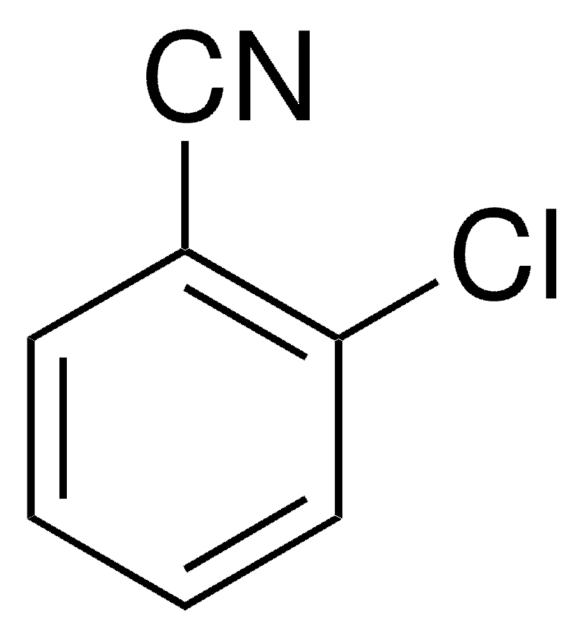
SMILES 字串
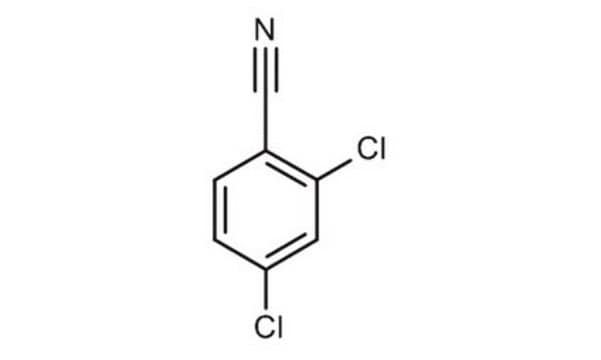
Clc1cccc(Cl)c1C#N
InChI
1S/C7H3Cl2N/c8-6-2-1-3-7(9)5(6)4-10/h1-3H
InChI 密鑰
YOYAIZYFCNQIRF-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
應用
2,6-Dichlorobenzonitrile can be used as a starting material to synthesize:
- 2,6-Dichlorobenzaldehyde using lithium N, N′-dimethylethylenediaminoaluminum hydride as a reducing agent.
- 5-(2,6-Dichlorophenyl)-2H-tetrazole via gold-catalyzed nucleophilic (3 + 2) cycloaddition reaction with sodium azide.
- 2,6-Dichlorobenzamide via hydrolysis using potassium tert-butoxide as a catalyst.
- Chloro-aminoindazole by reacting with hydrazine monohydrate.
- 2,6-Dichlorobenzenecarboselenoamide by treating with Woollins′ reagent.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Dermal - Aquatic Chronic 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
Synthetic application of gold nanoparticles and auric chloride for the synthesis of 5-substituted 1 H-tetrazoles
Kumar S, et al.
Royal Society of Chemistry Advances, 5(28), 21651-21658 (2015)
Selective conversion of aromatic nitriles to aldehydes by lithium N, N'-dimethylethylenediaminoaluminum hydride
Cha Jin-Soon, et al.
Bulletin of the Korean Chemical Society,, 23(12), 1697-1698 (2002)
Transition-metal-free hydration of nitriles using potassium tert-butoxide under anhydrous conditions
Midya GC, et al.
The Journal of Organic Chemistry, 80(8), 4148-4151 (2015)
The discovery and development of a safe, practical synthesis of ABT-869
Kruger AW, et al.
Organic Process Research & Development, 13(6), 1419-1425 (2009)
Synthesis of primary arylselenoamides by reaction of aryl nitriles with Woollins' reagent
Hua Guoxiong, et al.
Organic Letters, 8(23), 5251-5254 (2006)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门