推荐产品
方案
97%
折射率
n20/D 1.54 (lit.)
沸点
162 °C/20 mmHg (lit.)
密度
1 g/mL at 25 °C (lit.)
官能团
amine
phenyl
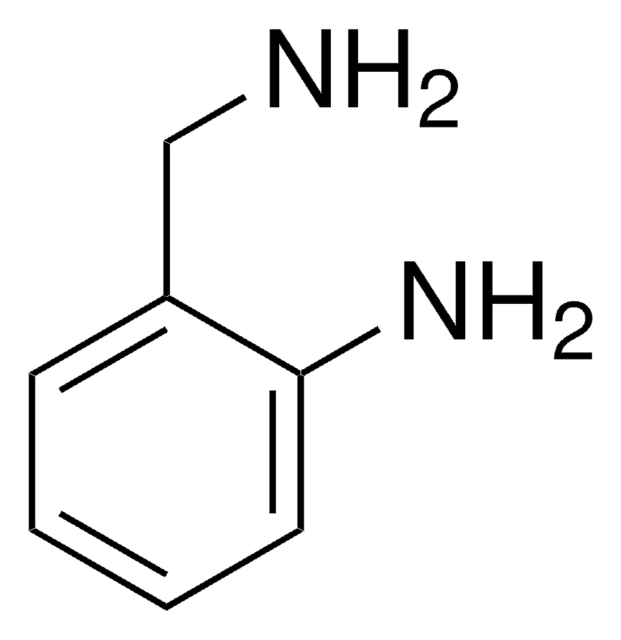
SMILES字符串
NCCNCc1ccccc1
InChI
1S/C9H14N2/c10-6-7-11-8-9-4-2-1-3-5-9/h1-5,11H,6-8,10H2
InChI key
ACYBVNYNIZTUIL-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般描述
N-Benzylethylenediamine participates in the one-pot synthesis of N,N,N′-trisubstituted guanidines. It undergoes condensation with dibenzoylmethane (1,3-diphenyl-1,3-propanedione) in stoichiometric ratio 1:1 to afford the corresponding Schiff monobase.
应用
N-Benzylethylenediamine may be used for the synthesis of N-benzyl-N,N′,N′-tris(tert-butyloxycarbonylmethyl)ethylenediamine and 3-benzyl-2-(phenyl-2-sulfonate)-2-imidazoline tetraheptylammonium salt.
警示用语:
Danger
危险声明
危险分类
Skin Corr. 1B
储存分类代码
8A - Combustible corrosive hazardous materials
WGK
WGK 3
闪点(°F)
203.0 °F - closed cup
闪点(°C)
95 °C - closed cup
个人防护装备
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
其他客户在看
A new method for the synthesis of tri-tert-butyl diethylenetriaminepentaacetic acid and its derivatives.
Achilefu S, et al.
The Journal of Organic Chemistry, 65(5), 1562-1565 (2000)
Anthony Weatherwax et al.
Organic letters, 7(16), 3461-3463 (2005-07-29)
Trans-disubstituted beta-lactams show increasing utility and prominence in numerous pharmaceutical applications, making their asymmetric synthesis an attractive goal for chemists. We introduce an anionic, nucleophilic catalyst system that provides an efficient, diastereoselective route to trans-disubstituted beta-lactams, a complement to our
Nickel (II) Complexes of Dibenzoylmethane and N-benzylethylenediamine, and Their Schiff Monobase.
Gutierrez JA, et al.
Journal of Coordination Chemistry, 28(3-4), 305-312 (1993)
Application of a-chloroaldoxime O-methanesulfonates to one-pot synthesis of N, N', N?-substituted guanidines via Tiemann rearrangement.
Yamamoto Y, et al.
Tetrahedron Letters, 50(42), 5813-5815 (2009)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门